
Tumor-Informed Minimal Residual Disease (MRD) Tests Market Report 2026
Global Outlook – By Technology (Next-Generation Sequencing, Polymerase Chain Reaction (PCR)-based, Other Technologies), By Product Type (Assay Kits, Instruments, Software, Services), By Sample Type (Peripheral Blood (Ctdna), Bone Marrow, Tissue Biopsy, Other Body Fluids), By Application (Colorectal Cancer, Lung Cancer, Breast Cancer, Hematological Malignancies, Other Applications), By End-User (Hospitals, Diagnostic Laboratories, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Tumor-Informed Minimal Residual Disease (MRD) Tests Market Overview
• Tumor-Informed Minimal Residual Disease (MRD) Tests market size has reached to $1.14 billion in 2025 • Expected to grow to $2.37 billion in 2030 at a compound annual growth rate (CAGR) of 15.8% • Growth Driver: The Rise In Cancer Prevalence Driving The Growth Of The Market Due To Unhealthy Lifestyles • Market Trend: Innovative Genomic Assay Detects Ultra-Low Levels Of Cancer DNA For Early Recurrence Monitoring • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Tumor-Informed Minimal Residual Disease (MRD) Tests Market?
Tumor-informed minimal residual disease (MRD) tests are specialized diagnostic assays that detect tiny amounts of cancer deoxyribonucleic acid (DNA) remaining in a patient’s body after treatment by analyzing genetic mutations specific to that individual’s tumor. These tests help monitor disease recurrence, guide therapy decisions, and assess treatment effectiveness with high sensitivity. The main technologies of tumor-informed minimal residual disease (MRD) tests include next-generation sequencing, polymerase chain reaction (PCR)-based methods, and other technologies. Next-generation sequencing is a high-throughput method that detects low levels of residual tumor deoxyribonucleic acid with high sensitivity, enabling precise monitoring of minimal residual disease in patients. The various product types include assay kits, instruments, software, and services and are applied for sample types such as peripheral blood (cDNA), bone marrow, tissue biopsy, and other body fluids. The key applications of colorectal cancer, lung cancer, breast cancer, hematological malignancies, and other applications, while the end-use includes hospitals, diagnostic laboratories, and research institutes.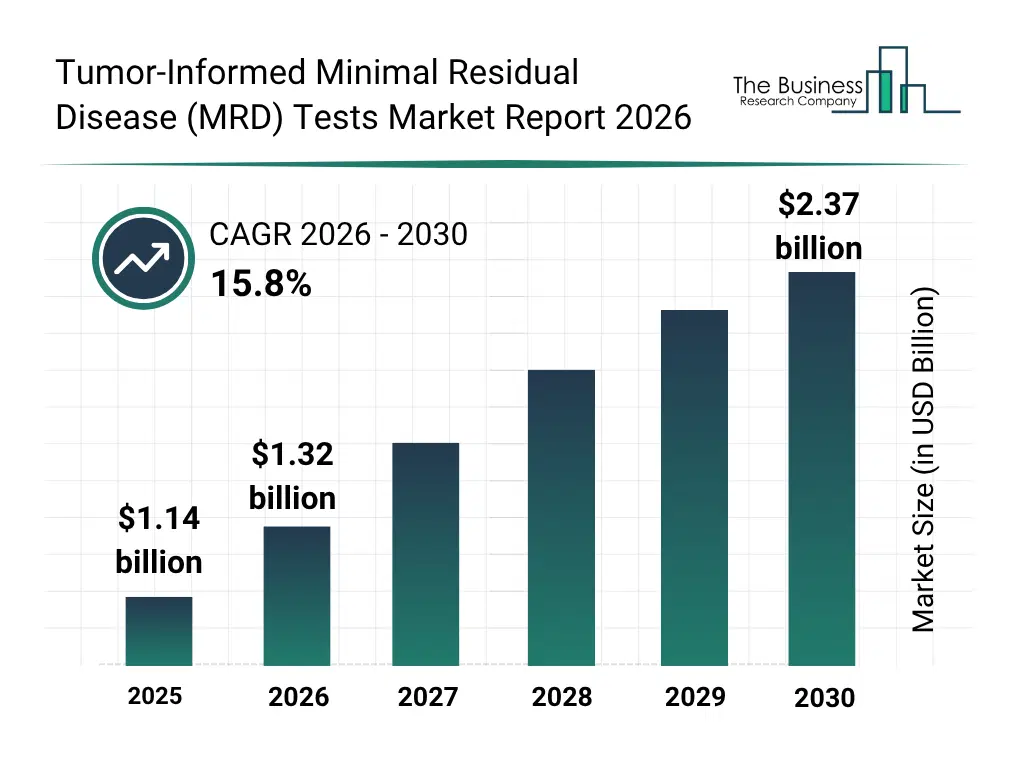
What Is The Tumor-Informed Minimal Residual Disease (MRD) Tests Market Size and Share 2026?
The tumor-informed minimal residual disease (MRD) tests market size has grown rapidly in recent years. It will grow from $1.14 billion in 2025 to $1.32 billion in 2026 at a compound annual growth rate (CAGR) of 15.6%. The growth in the historic period can be attributed to rising cancer prevalence, advancements in genetic sequencing, demand for post-treatment monitoring, regulatory approvals for molecular diagnostics, adoption of PCR and NGS technologies.What Is The Tumor-Informed Minimal Residual Disease (MRD) Tests Market Growth Forecast?
The tumor-informed minimal residual disease (MRD) tests market size is expected to see rapid growth in the next few years. It will grow to $2.37 billion in 2030 at a compound annual growth rate (CAGR) of 15.8%. The growth in the forecast period can be attributed to growth of personalized medicine, ai-driven data analytics for MRD, expansion of oncology research, development of non-invasive diagnostic assays, integration with hospital it systems. Major trends in the forecast period include personalized mrd monitoring, integration with clinical decision support systems, non-invasive sample analysis, high-sensitivity ctdna detection, predictive disease recurrence modeling.Global Tumor-Informed Minimal Residual Disease (MRD) Tests Market Segmentation
1) By Technology: Next-Generation Sequencing, Polymerase Chain Reaction (PCR)-based, Other Technologies 2) By Product Type: Assay Kits, Instruments, Software, Services 3) By Sample Type: Peripheral Blood (Ctdna), Bone Marrow, Tissue Biopsy, Other Body Fluids 4) By Application: Colorectal Cancer, Lung Cancer, Breast Cancer, Hematological Malignancies, Other Applications 5) By End-User: Hospitals, Diagnostic Laboratories, Research Institutes, Other End-Users Subsegments: 1) By Next-Generation Sequencing: Targeted Sequencing, Whole Genome Sequencing, Whole Exome Sequencing, Single Cell Sequencing, Other Sequencing Approaches 2) By Polymerase Chain Reaction (PCR)-Based: Digital Polymerase Chain Reaction, Real Time Polymerase Chain Reaction, Multiplex Polymerase Chain Reaction, Other Polymerase Chain Reaction Methods 3) By Other Technologies: Hybrid Capture Based, Bead Based Assays, Electrochemical Detection, Other Emerging TechnologiesWhat Is The Driver Of The Tumor-Informed Minimal Residual Disease (MRD) Tests Market?
The growing prevalence of cancer is expected to propel the growth of the tumor-informed minimal residual disease (MRD) tests market going forward. Cancer refers to a group of diseases characterized by uncontrolled growth and division of abnormal cells, which can invade surrounding tissues and spread to other parts of the body. The growing prevalence of cancer is due to unhealthy lifestyles, including poor diet, lack of physical activity, smoking, and excessive alcohol consumption, which increase the risk of developing various types of cancer. Tumor-informed minimal residual disease (MRD) tests help cancer management by detecting trace amounts of cancer-specific genetic material after treatment, enabling early identification of relapse and guiding personalized therapy decisions. For instance, in February 2024, according to the World Health Organization, a Switzerland-based international organization specializing in global health policy and public health initiatives, over 35 million new cancer cases are predicted in 2050, a 77% increase from the estimated 20 million cases in 2022. Therefore, the growing prevalence of cancer is driving the rapid growth of the tumor-informed minimal residual disease (MRD) tests industry.Key Players In The Global Tumor-Informed Minimal Residual Disease (MRD) Tests Market
Major companies operating in the tumor-informed minimal residual disease (MRD) tests market are Roche Diagnostics International Ltd., Thermo Fisher Scientific Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, Illumina Inc., Bio-Rad Laboratories Inc., Exact Sciences Corporation, QIAGEN N.V., Integrated DNA Technologies Inc., Natera Inc., NeoGenomics Laboratories Inc., Veracyte Inc., Foundation Medicine Inc., Adaptive Biotechnologies Corporation, Genetron Holdings Limited, Burning Rock Biotech Co Ltd., Personalis Inc., Foresight Diagnostics Inc., Predicine Inc., Guardant Health Inc.Global Tumor-Informed Minimal Residual Disease (MRD) Tests Market Trends and Insights
Major companies operating in the tumor informed minimal residual disease (MRD) tests market are focusing on developing innovative solutions, such as tissue informed whole genome sequencing (WGS) MRD assays to detect ultra-low levels of circulating tumor DNA for early monitoring of cancer recurrence and treatment response. A tissue-informed whole genome sequencing (WGS) MRD assay is a test that uses a patient’s tumor tissue to identify specific genetic mutations and then monitors blood for those mutations to detect minimal residual disease at very low levels. For instance, in September 2025, Foundation Medicine, Inc., a US-based precision medicine company, launched the Tissue-informed WGS MRD test via the FlexOMx Lab. This innovative, tumor-informed whole-genome sequencing test for molecular residual disease is designed for research use across early- to late-stage cancers. It tracks hundreds to thousands of tumor-specific variants in circulating tumor DNA with high precision, allowing for the accurate detection of minimal residual disease even at very low tumor levels, while also providing comprehensive genomic profiling to enhance understanding of recurrence and treatment resistance in clinical studies.What Are Latest Mergers And Acquisitions In The Tumor-Informed Minimal Residual Disease (MRD) Tests Market?
In June 2023, Quest Diagnostics Incorporated, a US-based healthcare technology and diagnostic information services company, acquired Haystack Oncology for an undisclosed amount. With this acquisition, Quest aims to enhance its cancer diagnostics portfolio by integrating highly sensitive minimal residual disease (MRD) liquid biopsy technology to improve early detection and personalized cancer care capabilities. Haystack Oncology Inc. is a US-based company that offers Haystack MRD, a tumor-informed ctDNA test for detecting minimal residual disease in cancer patients.Regional Insights
North America was the largest region in the tumor-informed minimal residual disease (MRD) tests market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Tumor-Informed Minimal Residual Disease (MRD) Tests Market?
The tumor-informed minimal residual disease (MRD) tests market consists of revenues earned by entities by providing services such as post-treatment cancer monitoring, prognostic assessment, therapy response evaluation, personalized treatment planning, and clinical trial support. The market value includes the value of related goods sold by the service provider or included within the service offering. The tumor-informed minimal residual disease (MRD) tests market also includes sales of next-generation sequencing (Ngs) panels, blood collection kits, bioinformatics software, reference standards and controls, and tumor tissue sequencing panels. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Tumor-Informed Minimal Residual Disease (MRD) Tests Market Report 2026?
The tumor-informed minimal residual disease (mrd) tests market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the tumor-informed minimal residual disease (mrd) tests industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Tumor-Informed Minimal Residual Disease (MRD) Tests Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.32 billion |
| Revenue Forecast In 2035 | $2.37 billion |
| Growth Rate | CAGR of 15.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Product Type, Sample Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Roche Diagnostics International Ltd., Thermo Fisher Scientific Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, Illumina Inc., Bio-Rad Laboratories Inc., Exact Sciences Corporation, QIAGEN N.V., Integrated DNA Technologies Inc., Natera Inc., NeoGenomics Laboratories Inc., Veracyte Inc., Foundation Medicine Inc., Adaptive Biotechnologies Corporation, Genetron Holdings Limited, Burning Rock Biotech Co Ltd., Personalis Inc., Foresight Diagnostics Inc., Predicine Inc., Guardant Health Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
