
Sternal Closure Devices Market Report 2026
Global Outlook – By Product Type (Wire Cerclage, Plate And Screw Systems, Cable Systems, Clips, Other Product Types), By Material (Stainless Steel, Titanium, Polyether Ether Ketone (PEEK), Other Materials), By Procedure (Median Sternotomy, Hemisternotomy, Bilateral Thoracosternotomy), By End-User (Hospitals, Cardiac Centers, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Sternal Closure Devices Market Overview
• Sternal Closure Devices market size has reached to $2.01 billion in 2025 • Expected to grow to $2.9 billion in 2030 at a compound annual growth rate (CAGR) of 7.6% • Growth Driver: Surge In Increasing Prevalence Of Cardiovascular Diseases Fueling The Growth Of The Market Due To Rising Demand For Open-Heart Surgeries • Market Trend: Advanced Fixation Technologies Improve Post-Operative Chest Stability And Patient Recovery Following Cardiac Surgery • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Sternal Closure Devices Market?
Sternal closure devices are medical tools designed to bring the sternum (breastbone) back together and provide stability after it has been surgically separated during procedures such as open-heart surgery. Their main function is to ensure secure closure of the bone, support effective healing, maintain chest wall stability, and minimize the risk of complications such as sternal separation or postoperative infections. The main product types of sternal closure devices include wire cerclage, plate and screw systems, cable systems, clips, and other product types. Wire cerclage devices are surgical tools used to stabilize and close the sternum after cardiac or thoracic surgery, ensuring proper healing and reducing postoperative complications. They are made from materials such as stainless steel, titanium, polyether ether ketone (PEEK), and other materials. They are used in procedures including median sternotomy, hemisternotomy, and bilateral thoracosternotomy, while the end-use includes hospitals, cardiac centers, ambulatory surgical centers, and other end users.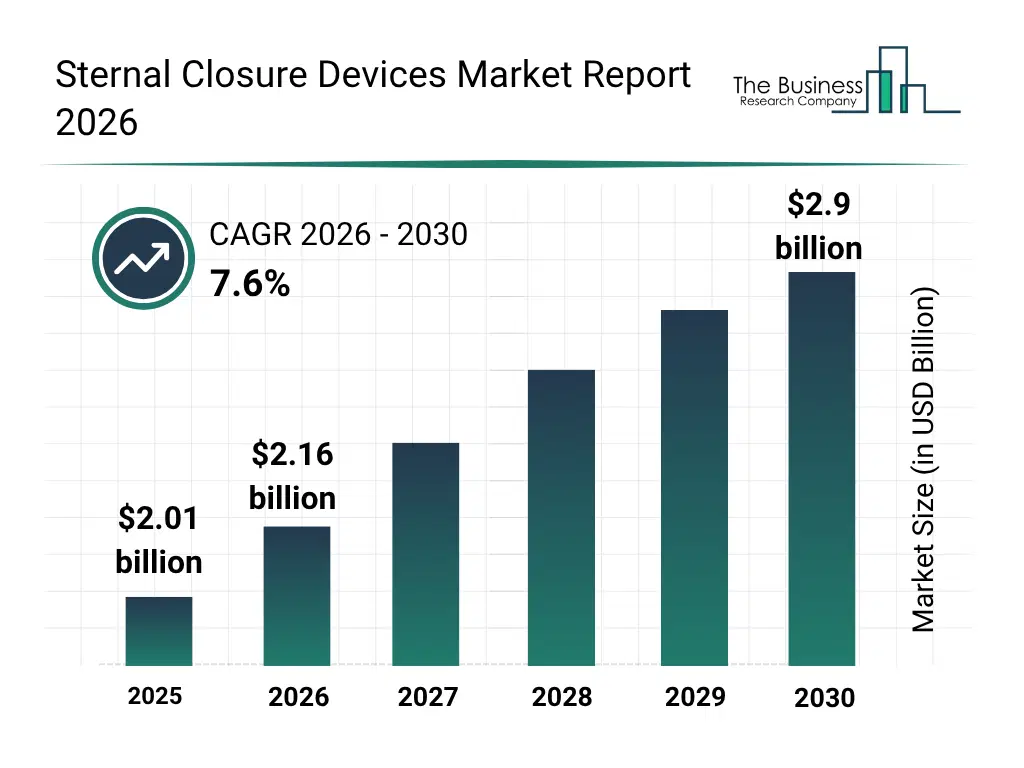
What Is The Sternal Closure Devices Market Size and Share 2026?
The sternal closure devices market size has grown strongly in recent years. It will grow from $2.01 billion in 2025 to $2.16 billion in 2026 at a compound annual growth rate (CAGR) of 7.4%. The growth in the historic period can be attributed to rising number of open-heart surgeries, adoption of traditional wire cerclage techniques, increasing awareness of postoperative complications, growth of cardiac care infrastructure, demand for reliable sternal stabilization.What Is The Sternal Closure Devices Market Growth Forecast?
The sternal closure devices market size is expected to see strong growth in the next few years. It will grow to $2.9 billion in 2030 at a compound annual growth rate (CAGR) of 7.6%. The growth in the forecast period can be attributed to innovation in bioabsorbable and hybrid closure systems, adoption of patient-specific titanium and peek devices, integration with digital surgical planning tools, demand for enhanced postoperative recovery monitoring, expansion of ambulatory surgical centers. Major trends in the forecast period include minimally invasive sternal closure techniques, patient-specific sternal fixation devices, advanced biocompatible materials, integration with surgical navigation systems, postoperative monitoring and analytics.Global Sternal Closure Devices Market Segmentation
1) By Product Type: Wire Cerclage, Plate And Screw Systems, Cable Systems, Clips, Other Product Types 2) By Material: Stainless Steel, Titanium, Polyether Ether Ketone (PEEK), Other Materials 3) By Procedure: Median Sternotomy, Hemisternotomy, Bilateral Thoracosternotomy 4) By End-User: Hospitals, Cardiac Centers, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Wire Cerclage: Stainless Steel Wires, Double Loop Wires, Figure Eight Wires, Twisted Wires, Coated Surgical Wires 2) By Plate And Screw Systems: Titanium Plates, Locking Plate Systems, Rigid Fixation Plates, Low Profile Plates, Self Tapping Screw Systems 3) By Cable Systems: Titanium Cables, Stainless Steel Cables, Adjustable Tension Cable Systems, Crimped Cable Systems, Multi Strand Cable Systems 4) By Clips: Titanium Sternal Clips, Stainless Steel Clips, Compression Clips, Locking Clips, Preloaded Clip Systems 5) By Other Product Types: Bioabsorbable Fixation Systems, Hybrid Closure Systems, Polymer Based Devices, Composite Fixation Devices, Reinforced Closure BandsWhat Are The Drivers Of The Sternal Closure Devices Market?
The increasing prevalence of cardiovascular diseases is expected to propel the growth of the sternal closure devices market going forward. Cardiovascular diseases refer to a group of disorders affecting the heart and blood vessels, including conditions such as coronary artery disease, heart failure, and stroke, which impair normal circulatory function. The prevalence of cardiovascular diseases is rising due to increasing exposure to unhealthy lifestyle patterns such as physical inactivity, which elevates cardiac risk and increases the number of patients requiring surgical intervention. Sternal closure devices secure the sternum following open-heart procedures, promoting proper bone healing, preserving chest stability, and minimizing risks of infection, instability, and delayed recovery to enhance overall postoperative outcomes in cardiovascular patients. For instance, in October 2024, according to the Centers for Disease Control and Prevention, a US-based government agency that monitors public health, cardiovascular disease deaths increased from 371,506 cases in 2022 to 919,032 deaths in 2023. Therefore, this increasing prevalence of cardiovascular diseases is driving the growth of the sternal closure devices industry. The rising cardiac surgery procedures are expected to propel the growth of the sternal closure devices market going forward. Cardiac surgery procedures refer to operative interventions performed on the heart and its surrounding structures to treat conditions such as coronary artery disease, valvular disorders, and congenital heart defects. The rising number of surgical procedures is primarily due to the increasing prevalence of chronic diseases associated with aging populations and lifestyle-related health conditions. The surge in cardiac surgical procedures directly increases the demand for sternal closure devices, as every open-heart surgery involving a median sternotomy requires secure and reliable sternal closure to facilitate healing and prevent postoperative complications. For instance, in March 2025, according to The Society of Thoracic Surgeons (STS) Adult Cardiac Surgery Database, a US-based clinical registry encompassing over 97% of cardiac surgery performed in the United States, overall cardiac surgical volumes increased by 4.2% in 2023 compared with 2022, reflecting a continued post-pandemic rebound in surgical activity across the country. Therefore, the rising cardiac surgery procedures are driving the growth of the sternal closure devices industryKey Players In The Global Sternal Closure Devices Market
Major companies operating in the sternal closure devices market are Stryker Corporation, B. Braun, Zimmer Biomet Holdings Inc., Arthrex Inc., Teleflex Incorporated, KLS Martin SE & Co. KG, Acumed LLC, Peters Surgical, DePuy Synthes, J.J.A.C.E. Ltd, Medicon eG, Kinamed Incorporated, ABYRX Inc., Jeil Medical Corporation, Able Medical Devices, Neos Surgery S.L., Praesidia S.R.L., MedXpert GmbH, Investigaciones y Desarrollos Argentinos S.R.L., Changzhou Waston Medical Appliance Co. Ltd., Dispomedica GmbH.Global Sternal Closure Devices Market Trends and Insights
Major companies operating in the sternal closure devices market are focusing on developing advanced rigid fixation solutions to enhance chest stability, reduce postoperative complications, and accelerate patient recovery after cardiac procedures. Rigid sternal fixation systems refer to implantable plating and screw-based devices that stabilize the sternum following median sternotomy, improving bone healing and minimizing the risk of sternal dehiscence. For instance, in August 2024, DePuy Synthes, a US-based medical device company, launched the MatrixSTERNUM Fixation System to provide enhanced sternal stability after open-heart surgery. The system features titanium plates and locking screw technology designed to deliver strong, reliable fixation, support bone healing, and reduce movement at the surgical site, thereby improving clinical outcomes and patient safety following cardiac surgery.Regional Insights
North America was the largest region in the sternal closure devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Sternal Closure Devices Market?
The sternal closure devices market consists of sales of sternal locking bands, bioresorbable sternal fixation devices, sternal bone adhesive systems, and titanium sternal reinforcement meshes. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Sternal Closure Devices Market Report 2026?
The sternal closure devices market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the sternal closure devices industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Sternal Closure Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.16 billion |
| Revenue Forecast In 2035 | $2.9 billion |
| Growth Rate | CAGR of 7.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Material, Procedure, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Stryker Corporation, B. Braun, Zimmer Biomet Holdings Inc., Arthrex Inc., Teleflex Incorporated, KLS Martin SE & Co. KG, Acumed LLC, Peters Surgical, DePuy Synthes, J.J.A.C.E. Ltd, Medicon eG, Kinamed Incorporated, ABYRX Inc., Jeil Medical Corporation, Able Medical Devices, Neos Surgery S.L., Praesidia S.R.L., MedXpert GmbH, Investigaciones y Desarrollos Argentinos S.R.L., Changzhou Waston Medical Appliance Co. Ltd., Dispomedica GmbH. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
