
Preclinical CRO Services Global Market Opportunities And Strategies To 2035
By Service Type (Target Validation And Screening, Drug Discovery And Drug ID, Preclinical Pharmacology, Toxicology And Safety Assessment, PK/ADME Studies, Biomarker Development, Other Key Service Stages), By Animal Model (Small Animal Model, Large Animal Model), By Assay Type (In-Vitro Bioassay, In-Vivo Efficacy Assay, Pharmacokinetic (PK) Assay, ADME Assay, Toxicology Assay, Other Assay Types), By Disease Area (Oncology, Central Nervous System (CNS) Disorders, Cardiovascular Diseases, Metabolic Disorders, Infectious Diseases, Autoimmune And Inflammatory Diseases, Other Disease Areas), And By Region, Opportunities And Strategies – Global Forecast To 2035
Preclinical CRO Services Market Definition
Preclinical contract research organization (CRO) services are outsourced research services that support the early-stage development of drugs, biologics, and medical devices before human clinical trials begin. The primary purpose of preclinical CRO services is to generate reliable scientific and safety data required for regulatory approval to proceed into clinical trials. They help pharmaceutical and biotechnology companies reduce development risk, control costs, accelerate timelines, and ensure compliance. The preclinical CRO services market consists of sales, by entities (organizations, sole traders, or partnerships) of services that are predominantly used by pharmaceutical companies, biotechnology firms, academic research institutions, and medical device manufacturers to evaluate the safety, efficacy, pharmacokinetics, and pharmacodynamics of candidate compounds. The services are typically utilized during the investigational new drug (IND)-enabling phase of a compound’s lifecycle, which occurs after initial target identification and lead optimization, but before first-in-human clinical studies.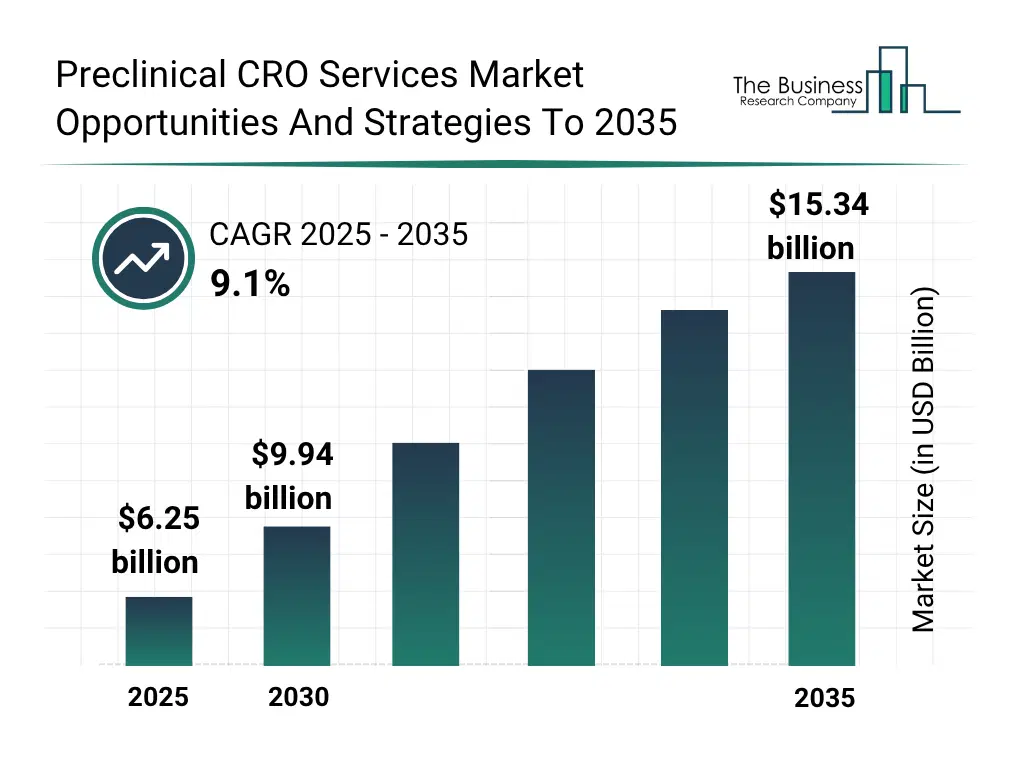
Preclinical CRO Services Market Size
The global preclinical CRO services market reached a value of nearly $6,246.1 million in 2025, having grown at a compound annual growth rate (CAGR) of 10.9% since 2020. The market is expected to grow from $ 6,246.1 million in 2025 to $9,943.5 million in 2030 at a rate of 9.7%. The market is then expected to grow at a CAGR of 9.1% from 2030 and reach $15,336.0 million in 2035. Growth in the historic period resulted from expansion of clinical trials and robust therapeutic pipeline, increased research and development (R&D) spending in life sciences, expansion of pharmaceutical and biotechnology industries and stringent regulatory and GLP compliance requirements. Factors that negatively affected growth in the historic period were high cost of preclinical studies and limited availability of skilled professionals. Going forward, expansion of biologics, cell & gene therapy pipelines, growing number of drug candidates requiring IND-enabling studies, growing demand to accelerate time-to-clinic and first-in-human studies and growing reliance on contract research organizations will drive growth. Factors that could hinder the growth of the preclinical CRO services market in the future include regulatory uncertainty and evolving global compliance standards, capacity constraints and skilled workforce shortages and impact of trade war and tariff.Preclinical CRO Services Market Segmentation
The preclinical CRO services market is segmented by service type, by animal model, by assay type and by disease area.By Type –
The preclinical CRO services market is segmented by type into:
- a) Target Validation And Screening
- b) Drug Discovery And Drug ID
- c) Preclinical Pharmacology
- d) Toxicology And Safety Assessment
- e) PK/ADME Studies
- f) Biomarker Development
- g) Other Key Service Stages
By Animal Model –
The preclinical CRO services market is segmented by animal model into:
- a) Small Animal Model
- b) Large Animal Model
By Assay Type –
The preclinical CRO services market is segmented by assay type into:
- a) In-Vitro Bioassay
- b) In-Vivo Efficacy Assay
- c) Pharmacokinetic (PK) Assay
- d) ADME Assay
- e) Toxicology Assay
- f) Other Assay Types
By Disease Area –
The preclinical CRO services market is segmented by disease area into:
- a) Oncology
- b) Central Nervous System (CNS) Disorders
- c) Cardiovascular Diseases
- d) Metabolic Disorders
- e) Infectious Diseases
- f) Autoimmune And Inflammatory Diseases
- g) Other Disease Areas
By Geography - The preclinical CRO services market is segmented by geography into:
- • China
- • India
- • Japan
- • Australia
- • Indonesia
- • South Korea
- • USA
- • France
- • Germany
- • UK
- • Italy
- • Spain
- • Russia
-
o Asia Pacific
