
Pharmaceutical Supply Chain Integrity Market Report 2026
Global Outlook – By Component (Software, Hardware, Services), By Technology (Serialization, Track And Trace Systems, Radio Frequency Identification Systems), By Deployment Mode (Cloud-Based Solutions, On-Premises Solutions), By Application (Manufacturers, Distributors, Wholesalers, Pharmacies, Hospitals, Other Applications), By End-User (Pharmaceutical Companies, Healthcare Providers, Logistics Providers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Pharmaceutical Supply Chain Integrity Market Overview
• Pharmaceutical Supply Chain Integrity market size has reached to $4.02 billion in 2025 • Expected to grow to $6.42 billion in 2030 at a compound annual growth rate (CAGR) of 9.9% • Growth Driver: Growing Digital Transformation Driving Growth In The Market Due To Real-Time Tracking, Blockchain Authentication, And Advanced Analytics • Market Trend: Advancements In Temperature-Controlled Warehousing Strengthen Pharmaceutical Supply Chain Integrity • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pharmaceutical Supply Chain Integrity Market?
Pharmaceutical supply chain integrity is the process of ensuring that medicines and healthcare products maintain their safety, authenticity, and quality at every stage of the supply chain, from procurement of raw materials and production to distribution and delivery to end users. It focuses on eliminating the risk of counterfeit, tampered, diverted, or inferior products entering the market. The main components of pharmaceutical supply chain integrity include software, hardware, and services. Software refers to platforms that help monitor, manage, and secure the pharmaceutical supply chain, ensuring product authenticity and compliance. These solutions incorporate technologies such as serialization, track and trace systems, and radio frequency identification systems and are deployed through cloud-based solutions and on-premises solutions. The various applications involved are manufacturers, distributors, wholesalers, pharmacies, hospitals, and other applications, and they are utilized by end users, including pharmaceutical companies, healthcare providers, logistics providers, and other end users.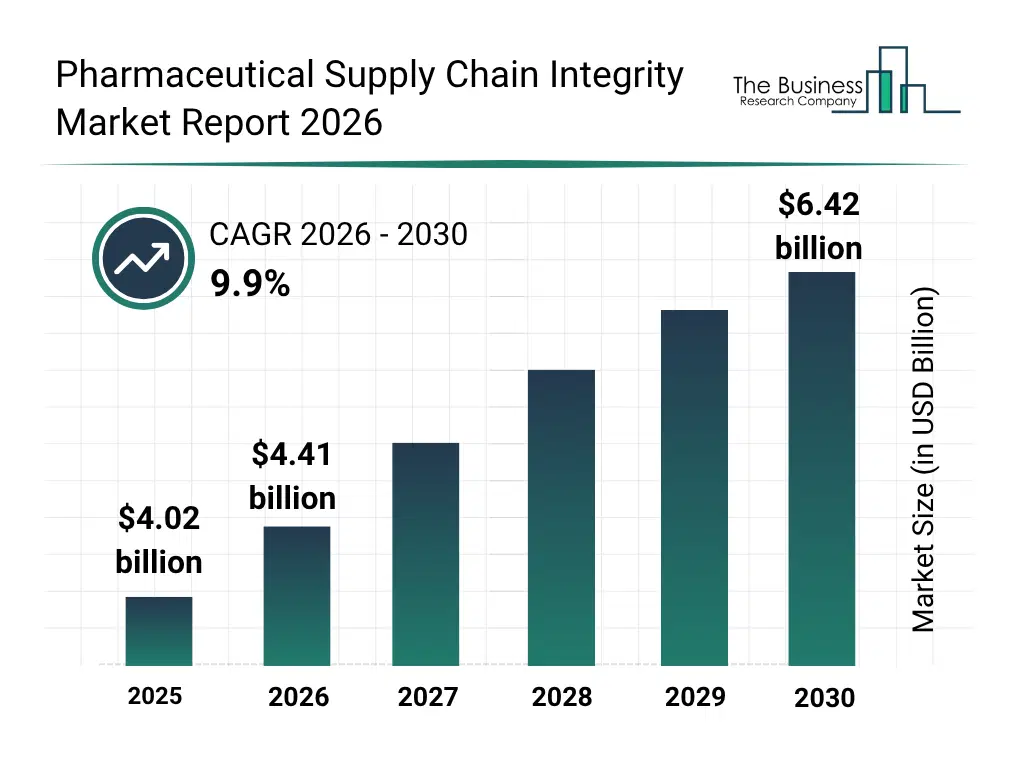
What Is The Pharmaceutical Supply Chain Integrity Market Size and Share 2026?
The pharmaceutical supply chain integrity market size has grown strongly in recent years. It will grow from $4.02 billion in 2025 to $4.41 billion in 2026 at a compound annual growth rate (CAGR) of 9.6%. The growth in the historic period can be attributed to rising counterfeit drug incidents, regulatory compliance mandates, growth of global pharmaceutical trade, adoption of barcode and serialization technology, demand for supply chain transparency.What Is The Pharmaceutical Supply Chain Integrity Market Growth Forecast?
The pharmaceutical supply chain integrity market size is expected to see strong growth in the next few years. It will grow to $6.42 billion in 2030 at a compound annual growth rate (CAGR) of 9.9%. The growth in the forecast period can be attributed to adoption of ai-powered monitoring systems, cloud-based track and trace implementation, expansion of healthcare and pharmaceutical logistics, integration of iot-enabled devices, predictive analytics for supply chain integrity. Major trends in the forecast period include real-time track and trace solutions, cold chain monitoring enhancement, automated serialization processes, integration with erp and scm systems, predictive risk management for supply chain.Global Pharmaceutical Supply Chain Integrity Market Segmentation
1) By Component: Software, Hardware, Services 2) By Technology: Serialization, Track And Trace Systems, Radio Frequency Identification Systems 3) By Deployment Mode: Cloud-Based Solutions, On-Premises Solutions 4) By Application: Manufacturers, Distributors, Wholesalers, Pharmacies, Hospitals, Other Applications 5) By End-User: Pharmaceutical Companies, Healthcare Providers, Logistics Providers, Other End Users Subsegments: 1) By Software: Track and Trace Software, Cold Chain Monitoring Software, Serialization Software 2) By Hardware: Barcode Scanners, Serialization Printers, Trackers And IoT Devices 3) By Services: Consulting Services, Maintenance And Support Services, Integration And Implementation ServicesWhat Is The Driver Of The Pharmaceutical Supply Chain Integrity Market?
The growing digital transformation is expected to propel the growth of the pharmaceutical supply chain integrity market going forward. Digital transformation refers to the integration of digital technologies into all aspects of a business to improve operations, enhance customer experiences, and drive innovation. Digital transformation is increasing because businesses need digital technologies to improve efficiency, respond quickly to market changes, and stay competitive. Digital transformation helps pharmaceutical supply chain integrity by enabling real-time tracking, blockchain-based authentication, and advanced analytics to prevent counterfeit drugs, ensure regulatory compliance, and maintain product quality throughout the supply chain. For instance, in November 2023, according to a report published by the Central Digital and Data Office, a UK-based government entity, the government’s focus on digital transformation has resulted in a 9% growth in the Government Digital and Data profession over the past six months, bringing the total number of professionals in this field to 28,337. Therefore, the growing digital transformation is driving the growth of the pharmaceutical supply chain integrity industry.Key Players In The Global Pharmaceutical Supply Chain Integrity Market
Major companies operating in the pharmaceutical supply chain integrity market are Avery Dennison Corporation, Zebra Technologies Corporation, Testo SE & Co. KGaA, Marchesini Group S.p.A., Uhlmann Pac-Systeme GmbH & Co. KG, WIPOTEC GmbH, Schreiner Group GmbH & Co. KG, Antares Vision S.p.A., Controlant hf., TraceLink Inc., Optel Group Inc., Tive Inc., Roambee Corporation, Digimarc Corporation, SEA Vision S.r.l., ParkourSC Inc., Aeologic Technologies Private Limited, Kezzler AS, ACVISS Technologies Private Limited, Ciphercode Technologies Private Limited, AlpVision SA, Covectra Inc., Veratrak Ltd., PharmaSecure Inc., and Tracekey Solutions GmbH.Global Pharmaceutical Supply Chain Integrity Market Trends and Insights
Major companies operating in the pharmaceutical supply chain integrity market are focusing on developing advanced solutions such as temperature-controlled warehousing to ensure drug safety, maintain product efficacy, and comply with regulatory standards throughout the supply chain. Temperature-controlled warehousing refers to storage facilities designed to maintain specific temperature ranges to preserve the quality, safety, and efficacy of temperature-sensitive pharmaceutical products throughout the supply chain. For instance, in August 2025, Celcius Logistics Solutions Private Limited, an India-based cold chain logistics company, launched Celcius+, a temperature-controlled dedicated pharma cold chain division, with an initial $0.006 billion (₹50 crore) investment. The initiative emphasizes GDP compliance, real-time monitoring via telematics and CCTV, AI-optimized routes, and a fleet of 100 reefer vehicles to minimize wastage and ensure supply chain integrity from manufacturer to end-user.What Are Latest Mergers And Acquisitions In The Pharmaceutical Supply Chain Integrity Market?
In February 2023, Overhaul Co., a US-based supply chain risk management and logistics technology company, acquired SensiGuard for an undisclosed amount. Through this acquisition, Overhaul aims to strengthen its life sciences supply chain integrity services by integrating SensiGuard’s advanced monitoring and compliance solutions, enabling improved tracking, temperature-sensitive handling, and regulatory adherence. SensiGuard is a US-based technology company specializing in supply chain monitoring and security solutions for life sciences and pharmaceutical products.Regional Insights
North America was the largest region in the pharmaceutical supply chain integrity market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pharmaceutical Supply Chain Integrity Market?
The pharmaceutical supply chain integrity market consists of revenues earned by entities by providing services such as authentication services, compliance monitoring, and supply chain data analytics. The market value includes the value of related goods sold by the service provider or included within the service offering. The pharmaceutical supply chain integrity market also includes sales of barcode scanners, RFID readers, tamper-evident packaging systems, and serialization and labeling machines. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Pharmaceutical Supply Chain Integrity Market Report 2026?
The pharmaceutical supply chain integrity market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pharmaceutical supply chain integrity industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pharmaceutical Supply Chain Integrity Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.41 billion |
| Revenue Forecast In 2035 | $6.42 billion |
| Growth Rate | CAGR of 9.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Technology, Deployment Mode, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Avery Dennison Corporation, Zebra Technologies Corporation, Testo SE & Co. KGaA, Marchesini Group S.p.A., Uhlmann Pac-Systeme GmbH & Co. KG, WIPOTEC GmbH, Schreiner Group GmbH & Co. KG, Antares Vision S.p.A., Controlant hf., TraceLink Inc., Optel Group Inc., Tive Inc., Roambee Corporation, Digimarc Corporation, SEA Vision S.r.l., ParkourSC Inc., Aeologic Technologies Private Limited, Kezzler AS, ACVISS Technologies Private Limited, Ciphercode Technologies Private Limited, AlpVision SA, Covectra Inc., Veratrak Ltd., PharmaSecure Inc., and Tracekey Solutions GmbH. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
