
Pharma ADMET Testing Global Market Opportunities And Strategies To 2035
By Testing Type (In Vivo ADMET Testing, In Vitro ADMET Testing, In Silico ADMET Testing), By Technology (Cell Culture, High Throughput, Molecular Imaging, OMICS Technology), By Application (Systemic Toxicity, Renal Toxicity, Hepatotoxicity, Neurotoxicity, Other Applications), And By Region, Opportunities And Strategies – Global Forecast To 2035
Pharma ADMET Testing Market Definition
Pharma ADMET testing refers to a group of laboratory and computational processes used in the pharmaceutical industry to evaluate the absorption, distribution, metabolism, excretion and toxicity (ADMET) profiles of drug candidates. It is an essential component of the drug discovery and development process, allowing researchers and developers to assess how a new compound behaves in the body and its potential safety risks before proceeding to clinical trials. The pharma ADMET testing market consists of sales, by entities (organizations, sole traders, or partnerships) is used by pharmaceutical companies, biotechnology firms, contract research organizations (CROs) and academic research institutions. The testing is conducted at early and preclinical stages of drug development and continues through the early phases of clinical trials to eliminate compounds with unfavorable pharmacokinetic or toxicity profiles.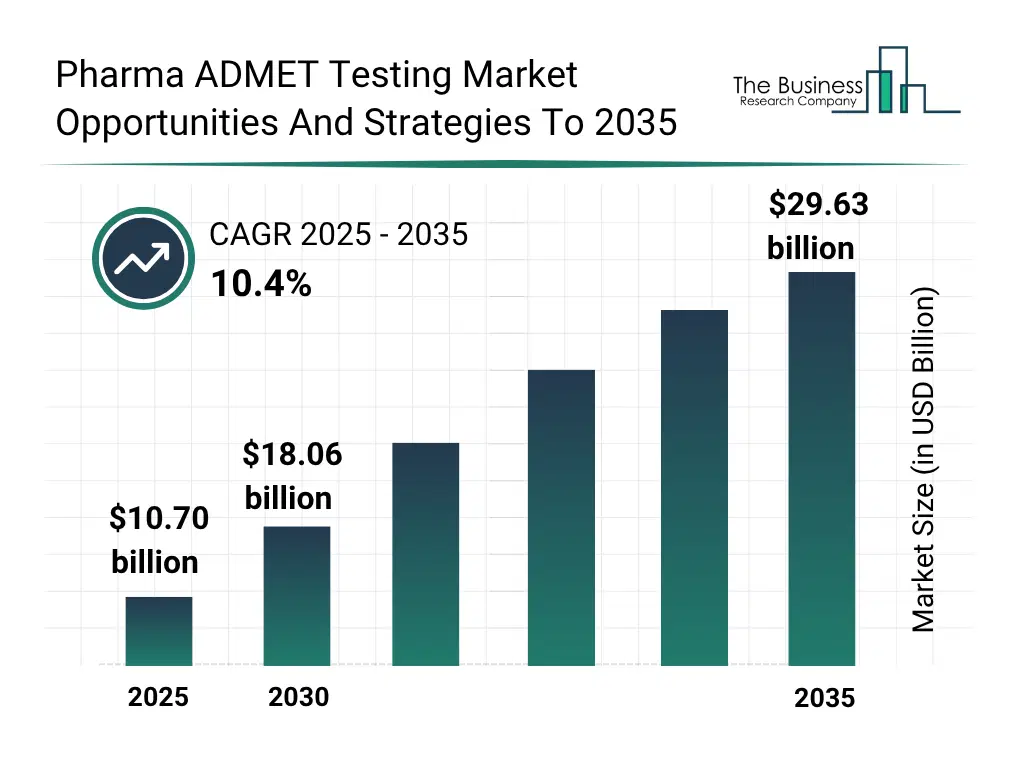
Pharma ADMET Testing Market Size
The global pharma ADMET testing market reached a value of nearly $10,697.9 million in 2025, having grown at a compound annual growth rate (CAGR) of 12.0% since 2020. The market is expected to grow from $10,697.9 million in 2025 to $18,062.9 million in 2030 at a rate of 11.0%. The market is then expected to grow at a CAGR of 10.4% from 2030 and reach $29,625.4 million in 2035. Growth in the historic period resulted from rising pharma-related R&D spending, growth in rare disease and orphan drug programs, expansion of combination therapy development and expansion of precision medicine and targeted therapies. Factors that negatively affected growth in the historic period were rising cost of in vivo animal studies and ethical restrictions and stringent regulatory requirements. Going forward, expansion of high-throughput screening (HTS) platforms, growth in multi-omics integration, rising public scrutiny of drug safety, shift toward complex biologics and novel modalities will drive growth. Factors that could hinder the growth of the pharma ADMET testing market in the future include limited high cost of preclinical testing and toxicology requirements, long duration of ADMET studies delays development timelines and impact of trade war and tariff.Pharma ADMET Testing Market Segmentation
The pharma ADMET testing market is segmented by testing type, by technology, and by application.By Testing Type –
The pharma ADMET testing market is segmented by testing type into:
- a) In Vivo ADMET Testing
- b) In Vitro ADMET Testing
- c) In Silico ADMET Testing
By Technology –
The pharma ADMET testing market is segmented by technology into:
- a) Cell Culture
- b) High Throughput
- c) Molecular Imaging
- d) OMICS Technology
By Application –
The pharma ADMET testing market is segmented by technology into:
- a) Systemic Toxicity
- b) Renal Toxicity
- c) Hepatotoxicity
- d) Neurotoxicity
- e) Other Applications
By Geography - The pharma ADMET testing market is segmented by geography into:
- • China
- • India
- • Japan
- • Australia
- • Indonesia
- • South Korea
- • USA
- • Canada
- • Brazil
- • France
- • Germany
- • UK
- • Italy
- • Spain
- • Russia
-
o Asia Pacific
o Africa
