
Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Report 2026
Global Outlook – By Product Type (Single Gene Panels, Multi Gene Panels, Comprehensive Genomic Panels), By Cancer Type (Lung Cancer, Breast Cancer, Colorectal Cancer, Pancreatic Cancer, Gastrointestinal Cancer, Hematological Malignancy), By Technology (Next Generation Sequencing, Polymerase Chain Reaction Based, Other Technologies), By Application (Oncology, Non Invasive Prenatal Testing, Transplantation, Other Applications), By End User (Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Overview
• Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels market size has reached to $1.27 billion in 2025 • Expected to grow to $2.78 billion in 2030 at a compound annual growth rate (CAGR) of 17% • Growth Driver: Surge In Prevalence Of Cancer Fueling The Growth Of The Market Due To Rising Demand For Early Detection And Continuous Monitoring • Market Trend: Innovations In Personalized Circulating Tumor DNA (ctDNA) Fingerprint Panels Enhance Ultra-Sensitive Molecular Residual Disease (MRD) Detection • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market?
Personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels are customized molecular diagnostic tests developed to identify and monitor tumor-specific genetic alterations present in circulating tumor DNA fragments in blood samples. These panels are created by analyzing an individual patient’s tumor tissue to determine unique mutation signatures and designing a tailored sequencing assay to track minimal residual disease, recurrence, or therapeutic response with high sensitivity. Personalized circulating tumor DNA (ctDNA) fingerprint panels utilize advanced next-generation sequencing and sophisticated bioinformatics tools enable highly accurate and non-invasive longitudinal cancer monitoring. The main product types of personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels include single gene panels, multi gene panels, and comprehensive genomic panels. Single gene panels are designed to identify specific tumor-related mutations in circulating tumor deoxyribonucleic acid to support precision oncology and disease monitoring. These panels are applied across cancer types including lung cancer, breast cancer, colorectal cancer, pancreatic cancer, gastrointestinal cancer, and hematological malignancy. They are developed using technologies such as next generation sequencing, polymerase chain reaction based methods, and other technologies. The various applications involved are oncology, non invasive prenatal testing, transplantation, and other applications, and they are used by several end users such as hospitals and clinics, diagnostic laboratories, research institutes, and other end users.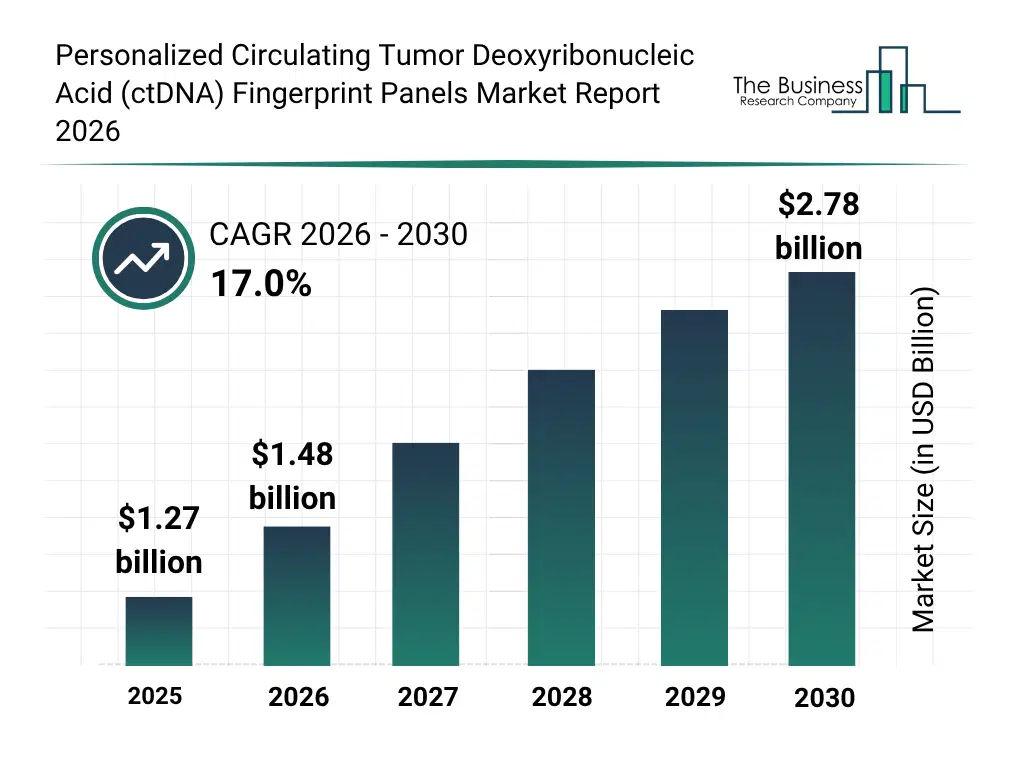
What Is The Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Size and Share 2026?
The personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market size has grown rapidly in recent years. It will grow from $1.27 billion in 2025 to $1.48 billion in 2026 at a compound annual growth rate (CAGR) of 16.7%. The growth in the historic period can be attributed to increasing cancer prevalence, advancements in next-generation sequencing, rising demand for non-invasive diagnostics, growing research in genomic biomarkers, regulatory support for precision medicine.What Is The Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Growth Forecast?
The personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market size is expected to see rapid growth in the next few years. It will grow to $2.78 billion in 2030 at a compound annual growth rate (CAGR) of 17.0%. The growth in the forecast period can be attributed to adoption of ai-powered genomic analysis, expansion of personalized oncology programs, integration with hospital information systems, increasing investments in ctDNA panels, demand for real-time treatment monitoring. Major trends in the forecast period include personalized ctdna assay development, longitudinal cancer monitoring, integration with clinical decision support systems, non-invasive disease tracking, ai-driven mutation analysis.Global Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Segmentation
1) By Product Type: Single Gene Panels, Multi Gene Panels, Comprehensive Genomic Panels 2) By Cancer Type: Lung Cancer, Breast Cancer, Colorectal Cancer, Pancreatic Cancer, Gastrointestinal Cancer, Hematological Malignancy 3) By Technology: Next Generation Sequencing, Polymerase Chain Reaction Based, Other Technologies 4) By Application: Oncology, Non Invasive Prenatal Testing, Transplantation, Other Applications 5) By End User: Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users Subsegments: 1) By Single Gene Panels: Tumor Specific Mutation Panels, Actionable Mutation Panels, Hereditary Cancer Panels 2) By Multi Gene Panels: Targeted Cancer Panels, Disease Specific Panels, Therapy Guidance Panels 3) By Comprehensive Genomic Panels: Pan Cancer Panels, Whole Exome Panels, Whole Genome PanelWhat Is The Driver Of The Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market?
The increasing prevalence of cancer is expected to propel the growth of the personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market going forward. Cancer refers to a collection of diseases marked by the uncontrolled proliferation of abnormal cells that can invade nearby tissues and spread to other parts of the body. The prevalence of cancer is rising due to longer life expectancy, improved early detection, and advances in treatment that allow patients to survive longer with the disease. Personalized circulating tumor DNA fingerprint panels support the cancer treatment by enabling early detection, continuous monitoring, and precise management of tumor progression, thereby improving survival outcomes and informing targeted treatment strategies. For instance, in October 2025, according to the American Cancer Society, a US-based nonprofit organization dedicated to cancer research and advocacy, there are about 18.6 million cancer survivors in the US, representing slightly more than 5% of the population, with projections estimating growth to 22 million by 2035. Therefore, the increasing prevalence of cancer is driving the growth of the personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels industry.Key Players In The Global Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market
Major companies operating in the personalized circulating tumor deoxyribonucleic acid (ctdna) fingerprint panels market are F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Laboratory Corporation of America Holdings, Agilent Technologies Inc., Illumina Inc., Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Natera Inc., Myriad Genetics Inc., NeoGenomics Laboratories Inc., Guardant Health Inc., Pacific Biosciences of California Inc., Burning Rock Biotech Limited, Personalis Inc., Biodesix Inc., OneCell Diagnostics Pvt. Ltd., CD Genomics, Creative Biolabs Inc., Predicine Inc., BioCreatrix Pte. Ltd., OncoDNA SA, Lucence Diagnostics Pte Ltd., Singlera Genomics Inc., Nonacus Ltd.Global Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Trends and Insights
Major companies operating in the personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market are focusing on developing ultra-sensitive, tumor-informed genome-based assays to improve early cancer detection, monitor treatment response, and detect molecular residual disease (MRD) with higher precision. Ultra-sensitive, tumor-informed, genome-based assays are advanced molecular diagnostics that identify and track patient-specific tumor mutations to detect extremely low levels of circulating tumor DNA (ctDNA) in blood. For instance, in April 2025, Natera Inc., a US-based molecular diagnostics company, announced the broad clinical launch of its ultra-sensitive Signatera Genome MRD test. The test leverages whole-genome sequencing (WGS) to design highly individualized ctDNA fingerprint panels tailored to each patient’s tumor profile, significantly increasing the number of tracked tumor variants and enhancing detection sensitivity at ultra-low ctDNA levels. This advancement strengthens MRD monitoring capabilities across multiple cancer types and supports earlier intervention and more informed treatment decision-making.What Are Latest Mergers And Acquisitions In The Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market?
In December 2025, Natera, Inc., a US-based provider of cell-free DNA testing and precision medicine solutions, acquired Foresight Diagnostics, Inc. for an undisclosed amount. With this acquisition, Natera aims to enhance its personalized ctDNA molecular residual disease testing capabilities, expand into lymphoma applications, and integrate Foresight's ultrasensitive phased variant technology into its Signatera platform. Foresight Diagnostics, Inc. is a US-based cancer diagnostics company and CLIA-registered laboratory that provides circulating tumor DNA–based molecular residual disease tests leveraging patented phased-seq technology.Regional Insights
North America was the largest region in the personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market?
The personalized circulating tumor deoxyribonucleic acid (ctDNA) fingerprint panels market consists of revenues earned by entities by providing services such as clinical data interpretation, laboratory testing services, and reporting and data management. The market value includes the value of related goods sold by the service provider or included within the service offering. The personalized circulating tumor DNA (ctDNA) fingerprint panels market also includes sales of sequencing reagents, PCR reagents, microfluidic cartridges, and benchtop sequencers. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Report 2026?
The personalized circulating tumor deoxyribonucleic acid (ctdna) fingerprint panels market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the personalized circulating tumor deoxyribonucleic acid (ctdna) fingerprint panels industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Personalized Circulating Tumor Deoxyribonucleic Acid (ctDNA) Fingerprint Panels Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.48 billion |
| Revenue Forecast In 2035 | $2.78 billion |
| Growth Rate | CAGR of 16.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Cancer Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Laboratory Corporation of America Holdings, Agilent Technologies Inc., Illumina Inc., Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Natera Inc., Myriad Genetics Inc., NeoGenomics Laboratories Inc., Guardant Health Inc., Pacific Biosciences of California Inc., Burning Rock Biotech Limited, Personalis Inc., Biodesix Inc., OneCell Diagnostics Pvt. Ltd., CD Genomics, Creative Biolabs Inc., Predicine Inc., BioCreatrix Pte. Ltd., OncoDNA SA, Lucence Diagnostics Pte Ltd., Singlera Genomics Inc., Nonacus Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
