
Neurointerventional Balloon Dilatation Catheter Market Report 2026
Global Outlook – By Product Type (Compliant, Non Compliant, Semi Compliant), By Balloon Shape (Standard, Steerable, Tapered), By Coating Technology (Drug Coated, Plain), By Application (Extracranial, Intracranial), By End-User (Ambulatory Surgical Centers, Hospitals, Specialty Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Neurointerventional Balloon Dilatation Catheter Market Overview
• Neurointerventional Balloon Dilatation Catheter market size has reached to $1.75 billion in 2025 • Expected to grow to $3.96 billion in 2030 at a compound annual growth rate (CAGR) of 17.8% • Growth Driver: Surge In Stroke And Intracranial Stenosis Fueling The Growth Of The Market Due To Aging Populations And Increased Risk Of Cerebrovascular Events • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Neurointerventional Balloon Dilatation Catheter Market?
A neurointerventional balloon dilatation catheter is a specialized medical device used to treat narrowed or blocked blood vessels in the brain. It features an inflatable balloon at its tip that, when positioned at the target site, expands to widen the vessel and restore proper blood flow. The catheter is designed for precise navigation through cerebral vasculature, minimizing trauma to surrounding tissues during neurovascular procedures. The main product types of neurointerventional balloon dilatation catheter are compliant, non-compliant, and semi-compliant. Compliant refers to catheters that can expand and conform to the vessel size, providing flexibility and safe navigation during neurointerventional procedures. These catheters are available in various balloon shapes, including standard, steerable, and tapered, and use different coating technologies, such as drug coated and plain. They are applied in extracranial and intracranial procedures and are used by end users such as ambulatory surgical centers, hospitals, and specialty clinics.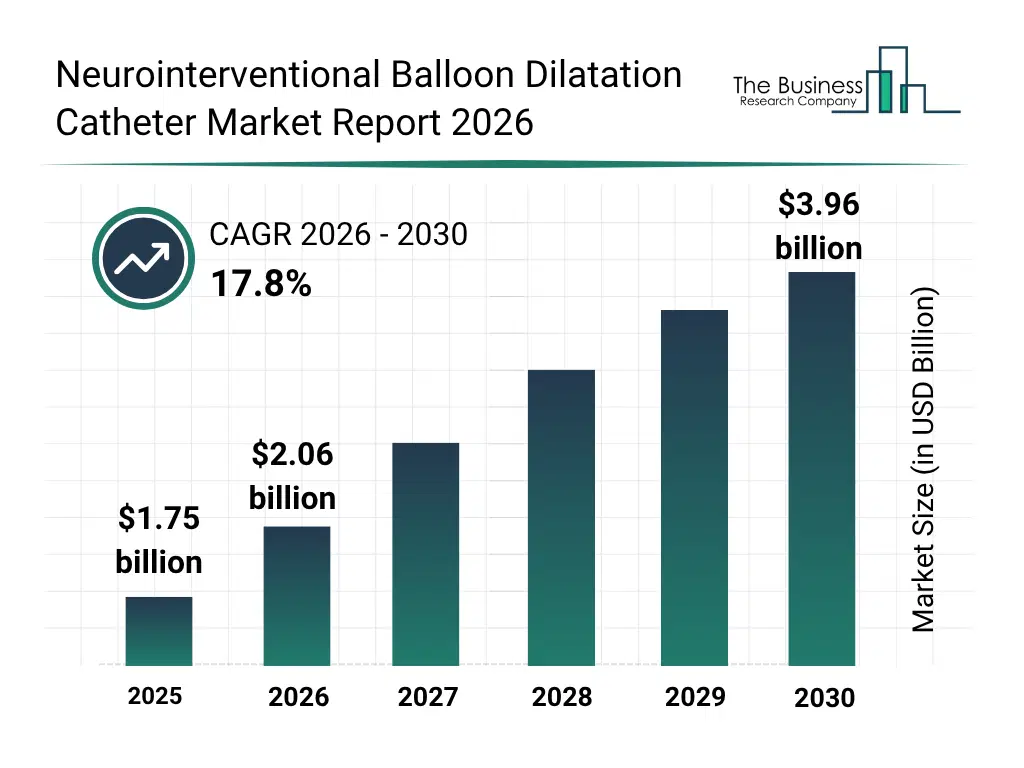
What Is The Neurointerventional Balloon Dilatation Catheter Market Size and Share 2026?
The neurointerventional balloon dilatation catheter market size has grown rapidly in recent years. It will grow from $1.75 billion in 2025 to $2.06 billion in 2026 at a compound annual growth rate (CAGR) of 17.5%. The growth in the historic period can be attributed to increasing prevalence of cerebrovascular diseases, advancements in catheter design and materials, rising number of neurovascular surgeries, growth of hospital and specialty clinic infrastructure, increasing awareness of minimally invasive procedures.What Is The Neurointerventional Balloon Dilatation Catheter Market Growth Forecast?
The neurointerventional balloon dilatation catheter market size is expected to see rapid growth in the next few years. It will grow to $3.96 billion by 2030 at a compound annual growth rate (CAGR) of 17.8%. The growth in the forecast period can be attributed to innovation in compliant and non-compliant balloon technologies, increasing adoption of drug-coated balloons, growth in ambulatory surgical centers, integration with AI-assisted imaging systems, expansion of intracranial treatment applications. Major trends in the forecast period include rising adoption of neurointerventional procedures, increasing use of drug-coated balloon technologies, growing preference for steerable and tapered balloons, expansion of intracranial and extracranial applications, integration of imaging and navigation systems in balloon catheters.Global Neurointerventional Balloon Dilatation Catheter Market Segmentation
1) By Product Type: Compliant, Non Compliant, Semi Compliant 2) By Balloon Shape: Standard, Steerable, Tapered 3) By Coating Technology: Drug Coated, Plain 4) By Application: Extracranial, Intracranial 5) By End-User: Ambulatory Surgical Centers, Hospitals, Specialty Clinics Subsegments: 1) By Compliant: Low Pressure Compliant Balloon, Medium Pressure Compliant Balloon, High Elasticity Compliant Balloon 2) By Non Compliant: High Pressure Non Compliant Balloon, Ultra High Pressure Non Compliant Balloon, Controlled Diameter Non Compliant Balloon 3) By Semi Compliant: Moderate Pressure Semi Compliant Balloon, Balanced Compliance Balloon, Flexible Semi Compliant BalloonWhat Are The Drivers Of The Neurointerventional Balloon Dilatation Catheter Market?
The rising prevalence of stroke and intracranial stenosis is expected to propel the growth of the neurointerventional balloon dilatation catheter market going forward. Stroke is often associated with intracranial stenosis, a narrowing of the brain’s arteries that increases the risk of ischemic events. The rising prevalence of stroke and intracranial stenosis is driven by aging populations, as older adults are more susceptible to arterial narrowing and cerebrovascular events. Neurointerventional balloon dilatation catheter helps treat stroke and intracranial stenosis by widening narrowed brain arteries, restoring blood flow, and reducing the risk of ischemic events. For instance, in December 2025, according to the Stroke Foundation, an Australia-based Non-profit organization, that stroke is among Australia’s top five killers, claiming 9,147 lives in 2024, with one person experiencing a stroke every 11 minutes. Therefore, the rising prevalence of stroke and intracranial stenosis is expected to drive the growth of the neurointerventional balloon dilatation catheter industry. The rising preference for minimally invasive procedures is expected to propel the growth of the neurointerventional balloon dilatation catheter market going forward. Minimally invasive procedures refer to medical or cosmetic treatments performed with minimal incisions or tissue damage, often using needles, catheters, or small instruments. The rising preference for minimally invasive procedures is driven by reduced risk of complications, as they involve smaller incisions and lower chances of infection or bleeding. Neurointerventional balloon dilatation catheter enables minimally invasive procedures by allowing precise widening of narrowed brain arteries through small vascular access, reducing the need for open surgery and lowering recovery time. For instance, in June 2024, according to the American Society of Plastic Surgeons, a US-based non-profit organization, minimally invasive procedures totaled 5.29 million in 2023, an 8% increase over 2022. Therefore, the rising preference for minimally invasive procedures is expected to drive the growth of the neurointerventional balloon dilatation catheter industry. The increasing healthcare expenditure is expected to propel the growth of the neurointerventional balloon dilatation catheter market going forward. Healthcare expenditure refers to the total amount of money spent by a country or organization on health services, medical goods, and public health programs. The increase in healthcare expenditure is driven by an aging population, as older adults require more medical care, treatments, and long-term health services. Neurointerventional balloon dilatation catheter helps optimize healthcare expenditure by enabling minimally invasive treatment of intracranial stenosis and stroke, reducing hospital stays, complications, and long-term care costs. For instance, in April 2025, according to the Office for National Statistics, a UK-based government department, healthcare expenditure was approximately £317 billion ($399.42 billion), growing 6.5% in nominal terms and 2.4% in real terms after adjusting for inflation. Therefore, the increasing healthcare expenditure is expected to drive the growth of the neurointerventional balloon dilatation catheter industry.Key Players In The Global Neurointerventional Balloon Dilatation Catheter Market
Major companies operating in the neurointerventional balloon dilatation catheter market are Johnson & Johnson, Medtronic plc, Stryker Corporation, Koninklijke Philips N.V., Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Kaneka Corporation, Nipro Corporation, Penumbra Inc., Lepu Medical Technology Co. Ltd., MicroPort Scientific Corporation, Asahi Intecc Co. Ltd., APT Medical Inc., Sino Medical Sciences Technology Inc., Peijia Medical Limited, Scientia Vascular Inc., Wallaby Medical Inc., Zylox-Tonbridge Medical Technology Co. Ltd., Acandis GmbH & Co. KG, Princeton Medical Scientific Inc., Cordis a Cardinal Health Company, DK MEDTECH Corporation.Regional Insights
North America was the largest region in the neurointerventional balloon dilatation catheter market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Neurointerventional Balloon Dilatation Catheter Market?
The neurointerventional balloon dilatation catheter market consists of sales of angiography contrast media, aspiration catheters, vascular access kits, and hemostatic devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Neurointerventional Balloon Dilatation Catheter Market Report 2026?
The neurointerventional balloon dilatation catheter market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the neurointerventional balloon dilatation catheter industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Neurointerventional Balloon Dilatation Catheter Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.06 billion |
| Revenue Forecast In 2035 | $3.96 billion |
| Growth Rate | CAGR of 17.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Balloon Shape, Coating Technology, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Medtronic plc, Stryker Corporation, Koninklijke Philips N.V., Boston Scientific Corporation, B. Braun Melsungen AG, Terumo Corporation, Kaneka Corporation, Nipro Corporation, Penumbra Inc., Lepu Medical Technology Co. Ltd., MicroPort Scientific Corporation, Asahi Intecc Co. Ltd., APT Medical Inc., Sino Medical Sciences Technology Inc., Peijia Medical Limited, Scientia Vascular Inc., Wallaby Medical Inc., Zylox-Tonbridge Medical Technology Co. Ltd., Acandis GmbH & Co. KG, Princeton Medical Scientific Inc., Cordis a Cardinal Health Company, DK MEDTECH Corporation. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
