
Nanobody-Based Diagnostics Market Report 2026
Global Outlook – By Technology (Immunoassays, Molecular Diagnostics, Biosensor-Based Detection, Fluorescence And Imaging Diagnostics), By Product Type (Reagents, Kits, Instruments, Other Product Types), By Application (Infectious Diseases, Oncology, Autoimmune Disorders, Cardiovascular Diseases, Other Applications), By End-User (Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Nanobody-Based Diagnostics Market Overview
• Nanobody-Based Diagnostics market size has reached to $1.13 billion in 2025 • Expected to grow to $2.07 billion in 2030 at a compound annual growth rate (CAGR) of 13% • Growth Driver: Rising Prevalence Of Infectious Diseases Driving The Market Growth Due To Increasing Need For Rapid And Accurate Diagnostic Solutions • Market Trend: Advancing Next-Generation Cancer Diagnostics And Therapies Through Innovative Theranostic Platforms • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Nanobody-Based Diagnostics Market?
Nanobody-based diagnostics utilize single-domain antibodies derived from camelids that are smaller, more stable, and highly specific compared to conventional antibodies. These nanobodies can efficiently bind to target molecules, enabling sensitive and rapid detection of biomolecules or pathogens. Their robustness and versatility make them valuable tools in various diagnostic assays and imaging applications. The main technologies of nanobody-based diagnostics include immunoassays, molecular diagnostics, biosensor-based detection, and fluorescence and imaging diagnostics. Immunoassays are laboratory techniques that detect or quantify specific molecules, such as proteins or hormones, using antibodies or antigens. The various product types include reagents, kits, instruments, and other product types. The key applications of infectious diseases, oncology, autoimmune disorders, cardiovascular diseases, and other applications, while the end-use includes hospitals and clinics, diagnostic laboratories, research institutes, and other end users.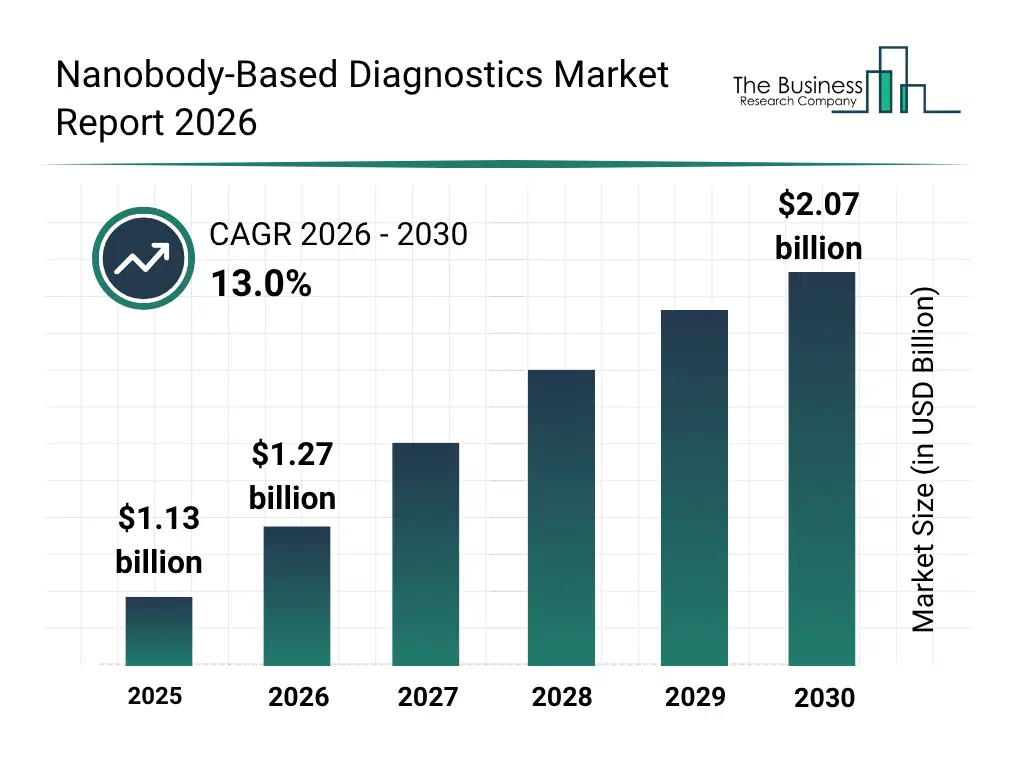
What Is The Nanobody-Based Diagnostics Market Size and Share 2026?
The nanobody-based diagnostics market size has grown rapidly in recent years. It will grow from $1.13 billion in 2025 to $1.27 billion in 2026 at a compound annual growth rate (CAGR) of 12.7%. The growth in the historic period can be attributed to advancements in antibody engineering technologies, increasing prevalence of infectious diseases, growth in cancer diagnostics demand, rising research funding in immunodiagnostics, expansion of rapid diagnostic testing platforms.What Is The Nanobody-Based Diagnostics Market Growth Forecast?
The nanobody-based diagnostics market size is expected to see rapid growth in the next few years. It will grow to $2.07 billion in 2030 at a compound annual growth rate (CAGR) of 13.0%. The growth in the forecast period can be attributed to growing demand for precision diagnostics, increasing integration of AI-based assay analysis tools, rising adoption of microfluidic biosensor systems, expansion of decentralized diagnostic testing, increasing focus on early disease detection biomarkers. Major trends in the forecast period include increasing adoption of lateral flow nanobody assays, rising demand for high-affinity single-domain antibody reagents, growing integration of nanobodies in point-of-care diagnostics, expansion of multiplexed biomarker detection platforms, rising use of nanobody-based imaging in disease monitoring.Global Nanobody-Based Diagnostics Market Segmentation
1) By Technology: Immunoassays, Molecular Diagnostics, Biosensor-Based Detection, Fluorescence And Imaging Diagnostics 2) By Product Type: Reagents, Kits, Instruments, Other Product Types 3) By Application: Infectious Diseases, Oncology, Autoimmune Disorders, Cardiovascular Diseases, Other Applications 4) By End-User: Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users Subsegments: 1) By Immunoassays: Enzyme Linked Immunosorbent Assay, Western Blotting, Lateral Flow Assay, Radioimmunoassay 2) By Molecular Diagnostics: Polymerase Chain Reaction Based Assays, Nucleic Acid Hybridization, Next Generation Sequencing, Isothermal Amplification Techniques 3) By Biosensor Based Detection: Electrochemical Biosensors, Optical Biosensors, Piezoelectric Biosensors, Microfluidic Biosensors 4) By Fluorescence and Imaging Diagnostics: Fluorescence Microscopy, Flow Cytometry, Fluorescence In Situ Hybridization, Molecular Imaging TechniquesWhat Are The Drivers Of The Nanobody-Based Diagnostics Market?
The rising prevalence of infectious diseases is expected to propel the growth of the nanobody based diagnostics market going forward. Infectious diseases refer to illnesses caused by harmful microorganisms, such as bacteria, viruses, fungi, or parasites, that can be transmitted from one person to another or through contact with contaminated environments. The prevalence of infectious diseases is growing due to closer human contact, which make it easier for pathogens to spread within communities. Nanobody‑based diagnostics provide rapid, highly specific, and sensitive detection of infectious agents, enabling early diagnosis and timely treatment, which helps control the spread of infectious diseases and improves patient outcomes. For instance, in March 2024, according to the Centers for Disease Control and Prevention, a US-based government agency, the number of tuberculosis cases rose from 8,320 in 2022 to 9,615 in 2023, an increase of 1,295 cases. Therefore, the rising prevalence of infectious diseases is driving the growth of the nanobody based diagnostics market. The increasing healthcare investments is expected to propel the growth of the nanobody based diagnostics market going forward. Healthcare investments refer to the allocation of financial resources by governments, organizations, and healthcare providers to improve medical infrastructure, services, technology, and overall healthcare delivery. Healthcare investments are rising due to the increasing demand for advanced medical services and modern technologies that improve patient care and health outcomes. Healthcare investments enhance the development and adoption of nanobody based diagnostics by funding advanced research, improving laboratory infrastructure, and enabling healthcare providers to implement cutting edge diagnostic technologies for more accurate and timely disease detection. For instance, in January 2026, according to the Centers for Medicare & Medicaid Services, a US-based government agency, in 2024, Medicare spending grew by 7.8%, reaching $1,118.0 billion and representing 21% of total national health expenditures (NHE), while Medicaid spending increased by 6.6% to $931.7 billion, accounting for 18% of total NHE. Therefore, the increasing healthcare investments is driving the growth of the nanobody based diagnostics market.Key Players In The Global Nanobody-Based Diagnostics Market
Major companies operating in the nanobody-based diagnostics market are Sanofi S.A., Thermo Fisher Scientific Inc., Merck KGaA, Agilent Technologies Inc., PerkinElmer Inc., Bruker Corporation, Bio-Rad Laboratories Inc., GenScript Biotech Corporation, Biocytogen LLC, Sino Biological Inc., Abzena plc, NanoTemper Technologies GmbH, RayBiotech Inc., Proteintech Group Inc., Creative Biolabs Inc., Isogenica Ltd., Beroni Group Limited, Immudex ApS, Cyclotek Pty Ltd, NanoTag Biotechnologies GmbH.Global Nanobody-Based Diagnostics Market Trends and Insights
Major companies operating in the nanobody-based diagnostics market are focusing on developing innovative solutions such as, advanced nanobody platforms to improve the accuracy and speed of disease detection. Advanced nanobody platforms use small, single-domain antibodies that are highly specific and stable, enabling faster tissue penetration, improved targeting of disease biomarkers, and enhanced diagnostic and therapeutic effectiveness compared to conventional antibodies. For instance, in September 2025, Cyclotek Pty Ltd, an Australia-based radiopharmaceutical company, was awarded a CRC-P Round 16 grant by the Australian Government to advance a nanobody-based theranostic platform for cancer. In collaboration with the University of Melbourne, Revela Bio Inc., and several local SMEs, the project focuses on developing targeted diagnostics and therapeutics that leverage nanobodies against the Fibroblast Activation Protein (FAP), a biomarker highly expressed in aggressive cancers such as lung, pancreatic, and ovarian cancer. Nanobodies combine the precision targeting of antibodies with the pharmacokinetics of small molecules, making them ideal for both diagnostic imaging and therapeutic applications. The initiative aims to develop advanced radiolabelling chemistry, conduct pre-clinical evaluations, and progress to first-in-human clinical trials, positioning Cyclotek Pty Ltd to scale and commercialize this innovative platform across Australia, New Zealand, and the broader Asia-Pacific region.Regional Insights
North America was the largest region in the nanobody-based diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Nanobody-Based Diagnostics Market?
The nanobody-based diagnostics market consists of revenues earned by entities by providing services such as biomarker detection, assay development, immunodiagnostic testing, molecular diagnostics support, and research-grade diagnostic solutions. The market value includes the value of related goods sold by the service provider or included within the service offering. The nanobody-based diagnostics market includes sales of nanobody-based assay kits, biosensors, diagnostic reagents, imaging probes, and targeted detection platforms. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Nanobody-Based Diagnostics Market Report 2026?
The nanobody-based diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the nanobody-based diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Nanobody-Based Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.27 billion |
| Revenue Forecast In 2035 | $2.07 billion |
| Growth Rate | CAGR of 12.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Product Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Sanofi S.A., Thermo Fisher Scientific Inc., Merck KGaA, Agilent Technologies Inc., PerkinElmer Inc., Bruker Corporation, Bio-Rad Laboratories Inc., GenScript Biotech Corporation, Biocytogen LLC, Sino Biological Inc., Abzena plc, NanoTemper Technologies GmbH, RayBiotech Inc., Proteintech Group Inc., Creative Biolabs Inc., Isogenica Ltd., Beroni Group Limited, Immudex ApS, Cyclotek Pty Ltd, NanoTag Biotechnologies GmbH. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
