
Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Report 2026
Global Outlook – By Product Type (Antimicrobial Urinary Catheters, Intermittent Catheters, Foley Catheters, Catheter Securement Devices, Urine Collection Bags), By Material Type (Silicone, Latex, Hydrogel Coated, Silver Alloy Coated), By Application Outlook (Urinary Incontinence Management, Acute Surgical Perioperative Care, Benign Prostatic Hyperplasia And Obstructive Urology, Neurogenic Bladder And Spinal Cord Injury, Other Applications), By End Use Outlook (Hospitals, Long Term Care Facilities, Home Care Setting, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Overview
• Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products market size has reached to $2.98 billion in 2025 • Expected to grow to $4.51 billion in 2030 at a compound annual growth rate (CAGR) of 8.7% • Growth Driver: The Rise In Hospital-Acquired Infections Driving The Growth Of The Market Due To Increasing Antimicrobial Resistance And Patient Risk • Market Trend: Innovative Female External Catheter Enhances Comfort And Reduces Infection Risk • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market?
Catheter-associated urinary tract infection (CAUTI) prevention urology products refer to medical devices and tools specifically designed to reduce the risk of urinary tract infections in patients requiring catheterization. These products focus on improving catheter management, maintaining urinary hygiene, and supporting infection control protocols in clinical settings. The main product types of catheter-associated urinary tract infection (CAUTI) prevention urology products include antimicrobial urinary catheters, intermittent catheters, foley catheters, catheter securement devices, and urine collection bags. Antimicrobial urinary catheters refer to catheters designed with antimicrobial coatings to prevent bacterial colonization and reduce the risk of infections. They are offered in silicone, latex, hydrogel coated, and silver alloy coated materials. The key applications of urinary incontinence management, acute surgical perioperative care, benign prostatic hyperplasia and obstructive urology, neurogenic bladder and spinal cord injury, and other applications, while the end-use includes hospitals, long term care facilities, home care settings, ambulatory surgical centers, and other end users.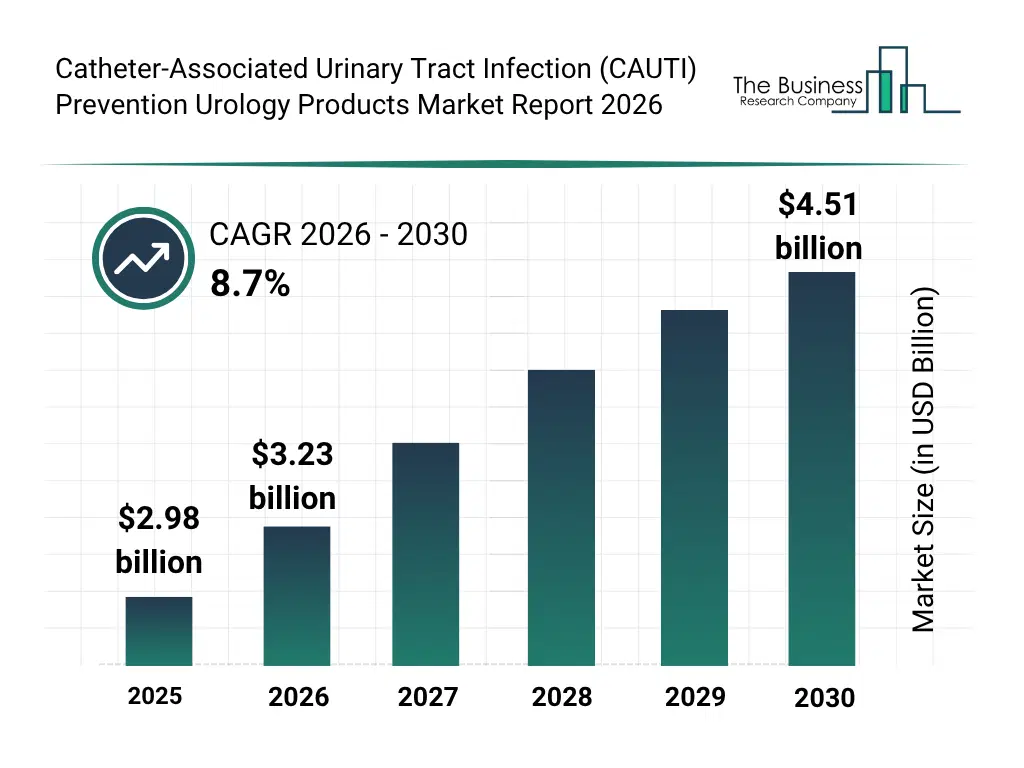
What Is The Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Size and Share 2026?
The catheter-associated urinary tract infection (CAUTI) prevention urology products market size has grown strongly in recent years. It will grow from $2.98 billion in 2025 to $3.23 billion in 2026 at a compound annual growth rate (CAGR) of 8.4%. The growth in the historic period can be attributed to increasing prevalence of catheter-associated urinary tract infections, growing hospital and long-term care facility patient population, adoption of infection control protocols, rising awareness of urinary hygiene, expansion of urology-focused medical devices.What Is The Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Growth Forecast?
The catheter-associated urinary tract infection (CAUTI) prevention urology products market size is expected to see strong growth in the next few years. It will grow to $4.51 billion in 2030 at a compound annual growth rate (CAGR) of 8.7%. The growth in the forecast period can be attributed to growing adoption of antimicrobial and smart catheters, increasing integration with patient monitoring systems, rising demand in home care settings, expansion of minimally invasive urology procedures, growing focus on regulatory compliance and infection prevention. Major trends in the forecast period include increasing adoption of antimicrobial urinary catheters, rising demand for closed system intermittent catheters, growing integration of catheter securement devices, expansion of smart foley catheters with monitoring features, rising focus on home care and long-term care catheter solutions.Global Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Segmentation
1) By Product Type: Antimicrobial Urinary Catheters, Intermittent Catheters, Foley Catheters, Catheter Securement Devices, Urine Collection Bags 2) By Material Type: Silicone, Latex, Hydrogel Coated, Silver Alloy Coated 3) By Application Outlook: Urinary Incontinence Management, Acute Surgical Perioperative Care, Benign Prostatic Hyperplasia And Obstructive Urology, Neurogenic Bladder And Spinal Cord Injury, Other Applications 4) By End Use Outlook: Hospitals, Long Term Care Facilities, Home Care Setting, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Antimicrobial Urinary Catheters: Silver Alloy Coated Urinary Catheters, Nitrofurazone Impregnated Urinary Catheters, Antibiotic Coated Urinary Catheters, Hydrophilic Coated Antimicrobial Urinary Catheters, Antiseptic Coated Urinary Catheters 2) By Intermittent Catheters: Hydrophilic Intermittent Catheters, Pre Lubricated Intermittent Catheters, Closed System Intermittent Catheters, Uncoated Intermittent Catheters, Single Use Sterile Intermittent Catheters 3) By Foley Catheters: Two Way Foley Catheters, Three Way Foley Catheters, Silicone Foley Catheters, Latex Foley Catheters, Temperature Sensing Foley Catheters 4) By Catheter Securement Devices: Adhesive Anchor Securement Devices, Leg Strap Securement Devices, Stat Lock Securement Devices, Stabilization Platform Devices, Adjustable Catheter Securement Systems 5) By Urine Collection Bags: Leg Mounted Urine Collection Bags, Bedside Drainage Urine Collection Bags, Closed System Drainage Bags, Anti Reflux Valve Urine Collection Bags, Graduated Measurement Urine Collection BagsWhat Is The Driver Of The Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market?
The rising hospital-acquired infection rates are expected to propel the growth of the catheter-associated urinary tract infection (CAUTI) prevention urology products market going forward. Hospital-acquired infection rates refer to the frequency of infections patients develop during hospital stays that were not present or incubating at the time of admission. The rising hospital-acquired infection rates are primarily due to antimicrobial resistance, which allows pathogens to survive standard treatments and spread more easily within healthcare settings. Catheter-associated urinary tract infection (CAUTI) prevention urology products help reduce hospital-acquired infection rates by minimizing bacterial colonization and biofilm formation on indwelling catheters, thereby lowering the risk of urinary tract infections in hospitalized patients. For instance, in May 2024, according to the Center for Infectious Disease Research and Policy (CIDRAP), a US-based disease research organization, the number of patients acquiring at least one healthcare-acquired infection in hospitals across the European Union and European Economic Area was nearly 4.3 million per year in 2022 and 2023. Therefore, rising hospital-acquired infection rates are driving the growth of the catheter-associated urinary tract infection (CAUTI) prevention urology products industry.Key Players In The Global Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market
Major companies operating in the catheter-associated urinary tract infection (CAUTI) prevention urology products market are Cardinal Health Inc., Medtronic plc, Medline Industries Inc., Stryker Corporation, Becton, Dickinson Company, Boston Scientific Corporation, B. Braun Melsungen AG, Olympus Corporation, Nipro Corporation, Coloplast A/S, Teleflex Incorporated, Cook Medical LLC, ConvaTec Group PLC, Hollister Incorporated, Merit Medical Systems Inc., Amsino International Inc., Laborie Medical Technologies Inc., Cathetrix Inc., Bactiguard AB, Poiesis Medical Inc.Global Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Trends and Insights
Major companies operating in the catheter associated urinary tract infection (CAUTI) prevention urology products market are focusing on developing innovative solutions, such as female external catheter devices, to enhance patient comfort, reduce infection risk, and improve overall urinary care outcomes. A female external catheter device is a non-invasive medical device worn externally over the female genital area to collect urine safely and comfortably, helping manage urinary incontinence and reduce infection risk. For instance, in January 2026, Wellspect HealthCare, a Sweden based medical device company, launched Surity Female External Catheter, an innovative non invasive urinary incontinence management product that supports women living with severe urinary incontinence and their caregivers. It is engineered to be easy to place, help prevent leakage, protect skin integrity, and support extended overnight use while integrating with compatible urine collection systems, addressing a significant gap in continence care where few options have existed. Its design emphasizes comfort, reliability, and predictable daily use in both home and healthcare settings as a contemporary alternative to indwelling catheters.What Are Latest Mergers And Acquisitions In The Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market?
In April 2024, Medline Industries LP, a US-based healthcare company, reached an agreement with Consure Medical to exclusively distribute the QiVi MEC male external urine management device in acute care settings across the United States. This agreement with Medline Industries LP aims to enhance patient care and reduce hospital-acquired infections by combining Medline’s extensive catheter and urology product portfolio with Consure Medical’s innovative external urine management technology. Consure Medical Inc. is a US-based medical device company specializing in providing urology products specifically designed for CAUTI (catheter-associated urinary tract infection) prevention.Regional Insights
North America was the largest region in the catheter-associated urinary tract infection (CAUTI) prevention urology products market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market?
The catheter-associated urinary tract infection (CAUTI) prevention urology products market consists of sales of antimicrobial‑coated urinary catheters, closed urine drainage systems, intermittent catheters, external urine management devices, and catheter hygiene products. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Report 2026?
The catheter-associated urinary tract infection (cauti) prevention urology products market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the catheter-associated urinary tract infection (cauti) prevention urology products industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Catheter-Associated Urinary Tract Infection (CAUTI) Prevention Urology Products Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.23 billion |
| Revenue Forecast In 2035 | $4.51 billion |
| Growth Rate | CAGR of 8.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Material Type, Application Outlook, End Use Outlook |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Cardinal Health Inc., Medtronic plc, Medline Industries Inc., Stryker Corporation, Becton, Dickinson Company, Boston Scientific Corporation, B. Braun Melsungen AG, Olympus Corporation, Nipro Corporation, Coloplast A/S, Teleflex Incorporated, Cook Medical LLC, ConvaTec Group PLC, Hollister Incorporated, Merit Medical Systems Inc., Amsino International Inc., Laborie Medical Technologies Inc., Cathetrix Inc., Bactiguard AB, Poiesis Medical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
