
Adeno-Associated Virus (AAV) Packaging Services Market Report 2026
Global Outlook – By Service Type (Research-Grade Adeno-Associated Virus (AAV) Packaging (RUO), Good Manufacturing Practice (GMP) Clinical Adeno-Associated Virus (AAV) Packaging, Good Manufacturing Practice (GMP) Late-Stage And Commercial Supply, Analytical And Release Testing, Process Development And Tech Transfer), By Vector Type (Adeno-Associated Virus (AAV) 2, Adeno-Associated Virus (AAV) 5, Adeno-Associated Virus (AAV) 8, Adeno-Associated Virus (AAV) 9), By End User (Biotechnology And Biopharmaceutical Companies, Academic And Research Institutes, Contract Research Organizations, Contract Manufacturing Organizations, Healthcare And Diagnostic Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Adeno-Associated Virus (AAV) Packaging Services Market Overview
• Adeno-Associated Virus (AAV) Packaging Services market size has reached to $1.48 billion in 2025 • Expected to grow to $2.52 billion in 2030 at a compound annual growth rate (CAGR) of 11.3% • Growth Driver: Rising Gene Therapy Development Driving Growth Of The Market Due To Increasing Prevalence Of Genetic And Rare Diseases • Market Trend: Advancements In Serotype-Specific Viral Vector Manufacturing Capabilities • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Adeno-Associated Virus (AAV) Packaging Services Market?
Adeno-associated virus (AAV) packaging services are specialized laboratory services that produce recombinant AAV vectors by inserting a target gene into AAV particles for research or therapeutic use. These services involve plasmid design, vector assembly, viral production in suitable host cells, purification, and quality control testing to ensure high titer and purity. They enable efficient and stable gene delivery for research and therapeutic applications. The main service types of adeno-associated virus (AAV) packaging services include research-grade adeno-associated virus (AAV) packaging (RUO), good manufacturing practice (GMP) clinical adeno-associated virus (AAV) packaging, good manufacturing practice (GMP) late-stage and commercial supply, analytical and release testing, and process development and tech transfer. Research-grade AAV packaging (RUO) refers to services designed for laboratory research and preclinical studies. The vector types include adeno-associated virus (AAV) 2, AAV 5, AAV 8, and AAV 9 with different scales of production, including small-scale production, mid-scale production, and large-scale production. The therapeutic applications include ophthalmology, neuromuscular and central nervous system disorders, metabolic or rare genetic disorders, oncology, cardiovascular, and others, serving end users such as biotechnology and biopharmaceutical companies, academic and research institutes, contract research organizations, contract manufacturing organizations, and healthcare and diagnostic laboratories.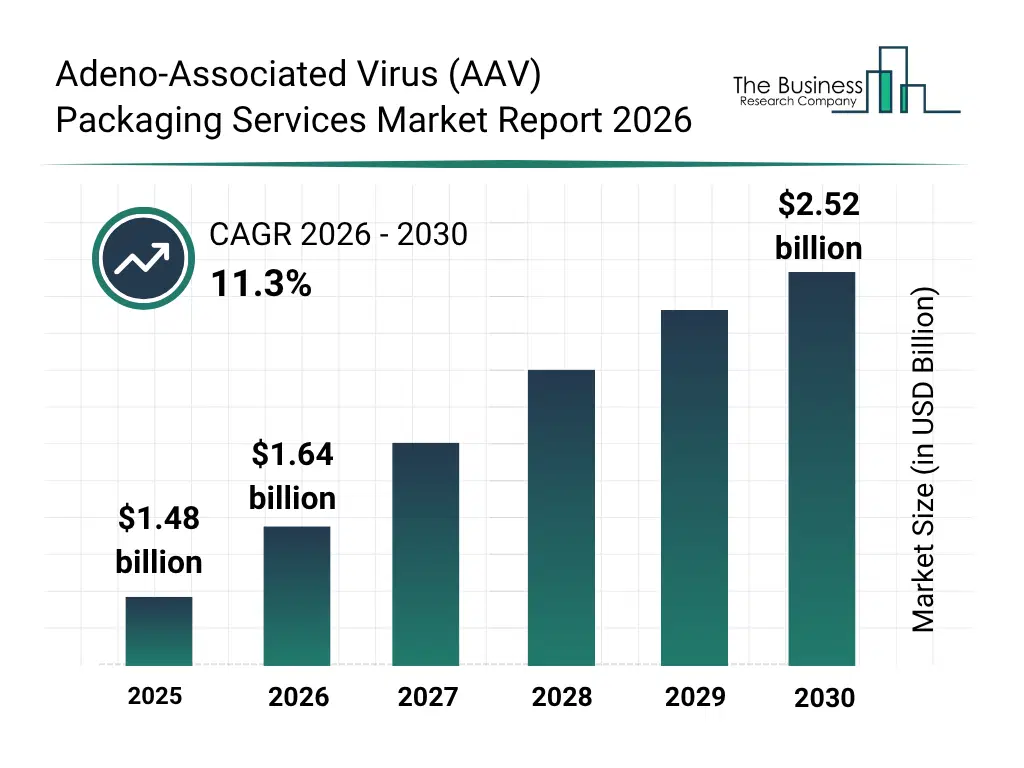
What Is The Adeno-Associated Virus (AAV) Packaging Services Market Size and Share 2026?
The adeno-associated virus (AAV) packaging services market size has grown rapidly in recent years. It will grow from $1.48 billion in 2025 to $1.64 billion in 2026 at a compound annual growth rate (CAGR) of 11.1%. The growth in the historic period can be attributed to rising gene therapy research activities, increasing funding for rare disease research, growth in preclinical and clinical trial pipelines, expansion of biotechnology startups, advancements in viral vector production technologies.What Is The Adeno-Associated Virus (AAV) Packaging Services Market Growth Forecast?
The adeno-associated virus (AAV) packaging services market size is expected to see rapid growth in the next few years. It will grow to $2.52 billion in 2030 at a compound annual growth rate (CAGR) of 11.3%. The growth in the forecast period can be attributed to increasing approvals of gene therapies, rising demand for commercial-scale GMP manufacturing, growing investments in precision medicine, expansion of cell and gene therapy pipelines, increasing strategic partnerships between biotech firms and cdmos. Major trends in the forecast period include increasing demand for high-titer and high-purity aav production, expansion of GMP-compliant clinical-grade aav packaging, growing focus on scalable large-scale manufacturing capabilities, rising adoption of advanced analytical and release testing services, increasing outsourcing of aav vector manufacturing to cdmos.Global Adeno-Associated Virus (AAV) Packaging Services Market Segmentation
1) By Service Type: Research-Grade Adeno-Associated Virus (AAV) Packaging (RUO), Good Manufacturing Practice (GMP) Clinical Adeno-Associated Virus (AAV) Packaging, Good Manufacturing Practice (GMP) Late-Stage And Commercial Supply, Analytical And Release Testing, Process Development And Tech Transfer 2) By Vector Type: Adeno-Associated Virus (AAV) 2, Adeno-Associated Virus (AAV) 5, Adeno-Associated Virus (AAV) 8, Adeno-Associated Virus (AAV) 9 3) By Scale Of Production: Small-Scale Production, Mid-Scale Production, Large-Scale Production 4) By Therapeutic Application: Ophthalmology, Neuromuscular And Central Nervous System, Metabolic Or Rare Genetic Disorders, Oncology, Cardiovascular, Other Indications 5) By End User: Biotechnology And Biopharmaceutical Companies, Academic And Research Institutes, Contract Research Organizations, Contract Manufacturing Organizations, Healthcare And Diagnostic Laboratories Subsegments: 1) By Research-Grade Adeno-Associated Virus (AAV) Packaging (RUO): Small Scale Production, Pilot Scale Production, Custom Vector Design, Preclinical Study Supply 2) By GMP Clinical Adeno-Associated Virus (AAV) Packaging: Phase One Clinical Supply, Phase Two Clinical Supply, Phase Three Clinical Supply, Patient Specific Batch Production 3) By GMP Late-Stage And Commercial Supply: Commercial Batch Production, Technology Transfer Support, Large Scale Manufacturing, Regulatory Submission Support 4) By Analytical And Release Testing: Potency Testing, Purity Testing, Identity Testing, Safety Testing, Stability Testing 5) By Process Development And Tech Transfer: Upstream Process Development, Downstream Process Development, Scale-Up Process Optimization, Technology Transfer To Manufacturing, Process CharacterizationWhat Is The Driver Of The Adeno-Associated Virus (AAV) Packaging Services Market?
The rising demand for gene therapy development is expected to propel the growth of the adeno-associated virus (AAV) packaging services market going forward. Gene therapy development refers to creating treatments that modify or replace faulty genes to treat diseases. Demand for gene therapy development is growing due to the rising prevalence of genetic and rare diseases, which is increasing the need for advanced treatments that target the underlying genetic causes of these conditions. Adeno-associated virus (AAV) packaging services enhance gene therapy development by providing specialized viral vector production and purification services that enable scalable, high-quality AAV supply for clinical research and therapeutic use. For instance, in July 2023, according to the American Society of Gene & Cell Therapy (ASGCT), a US-based professional membership organization, at the end of the first quarter of 2023, there were 247 gene therapies in Phase II, but this number rose by 5% to reach 260 by the end of the second quarter. Therefore, the rising demand for gene therapy development is driving the growth of the adeno-associated virus (AAV) packaging services industry.Key Players In The Global Adeno-Associated Virus (AAV) Packaging Services Market
Major companies operating in the adeno-associated virus (AAV) packaging services market are Thermo Fisher Scientific Inc., AGC Biologics Inc., Lonza Group AG, Charles River Laboratories International Inc., Resilience Biotechnologies Inc., GenScript Biotech Corporation, Takara Bio Inc., OxB CDMO Inc., 3PBIOVIAN, VectorBuilder, Genezen Labs, Addgene Inc., PackGene Biotech Inc., Porton Advanced Solutions Ltd., Creative Biogene, Yposkesi SAS, AAVnerGene Inc., Virovek Inc., WuXi Advanced Therapies Co. Ltd., BioInnovatise Inc.Global Adeno-Associated Virus (AAV) Packaging Services Market Trends and Insights
Major companies operating in the adeno-associated virus (AAV) packaging services market are focusing on developing innovative solutions, such as on-demand AAV viral vector packaging platforms, to enhance flexibility, accelerate research timelines, and ensure consistent high-quality viral vector production. On-demand AAV viral vector packaging platforms are services that provide customized AAV production in specific serotypes with quality controls, helping researchers save time and obtain consistent, high-titer vectors for gene delivery applications. For instance, in January 2025, Addgene Inc., a US-based nonprofit scientific organization, launched Packaged on Request, an innovative AAV packaging service. It allows scientists to choose from over 3,000 AAV plasmids in Addgene’s repository and request packaging in serotypes including AAV1, AAV2, AAV5, AAV8, and AAV Retrograde capsids. Each preparation undergoes comprehensive quality control procedures, including ddPCR titering, purity testing, sterility testing, and viral genome sequencing, ensuring research-grade standards consistent with in-stock viral preps. The service also streamlines legal approvals and technology transfer processes, reducing administrative burden while maintaining high titers suitable for diverse in vivo and in vitro applications.What Are Latest Mergers And Acquisitions In The Adeno-Associated Virus (AAV) Packaging Services Market?
In December 2023, Ajinomoto Co. Inc., a Japan-based food and biotechnology company, acquired Forge Biologics Holdings LLC for $0.54 billion (JPY 82.8 billion). With this acquisition, Ajinomoto aimed to integrate its proprietary aminoscience platform with Forge Biologics’ gene therapy CDMO capabilities to accelerate its expansion into advanced healthcare and gene therapy manufacturing markets. Forge Biologics Holdings LLC is a US-based gene therapy contract development and manufacturing organization (CDMO) that specializes in providing adeno-associated virus (AAV) production and plasmid DNA manufacturing and packaging for clinical and commercial development.Regional Insights
North America was the largest region in the adeno-associated virus (AAV) packaging services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Adeno-Associated Virus (AAV) Packaging Services Market?
The adeno-associated virus (AAV) packaging services market consists of revenues earned by entities by providing services such as vector design and cloning, plasmid construction, recombinant AAV production, viral packaging and amplification, purification, titration and quality control testing, and custom serotype selection. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Adeno-Associated Virus (AAV) Packaging Services Market Report 2026?
The adeno-associated virus (aav) packaging services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the adeno-associated virus (aav) packaging services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Adeno-Associated Virus (AAV) Packaging Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.64 billion |
| Revenue Forecast In 2035 | $2.52 billion |
| Growth Rate | CAGR of 11.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service Type, Vector Type, Scale Of Production, Therapeutic Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | Major companies operating in the adeno-associated virus (AAV) packaging services market are Thermo Fisher Scientific Inc., AGC Biologics Inc., Lonza Group AG, Charles River Laboratories International Inc., Resilience Biotechnologies Inc., GenScript Biotech Corporation, Takara Bio Inc., OxB CDMO Inc., 3PBIOVIAN, VectorBuilder, Genezen Labs, Addgene Inc., PackGene Biotech Inc., Porton Advanced Solutions Ltd., Creative Biogene, Yposkesi SAS, AAVnerGene Inc., Virovek Inc., WuXi Advanced Therapies Co. Ltd., BioInnovatise Inc. </b> |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
